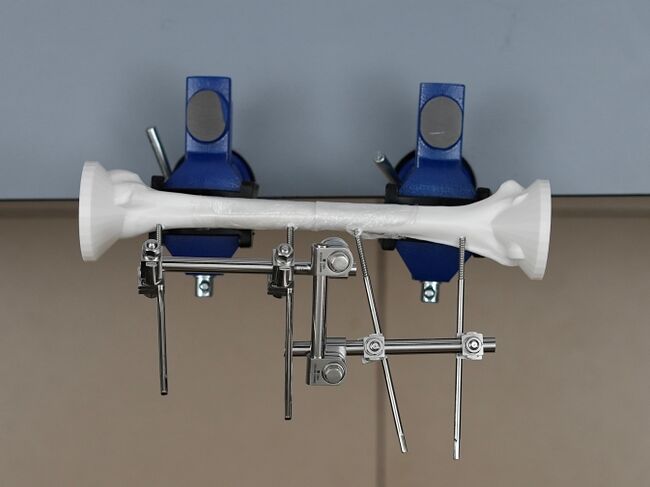
This module allows medical officers and surgeons who are not orthopedic specialists to become confident and competent in irrigation and debridement, powered and manual drilling, positioning and correctly inserting Schanz screws, and constructing the rod-to-rod modular frame as part of external fixation procedures for open humeral shaft fractures performed in regions without specialist coverage. To maximize patient safety, this module teaches learners to use a powered drill to insert self-drilling Schanz screws through the near cortex and then manually advance Schanz screws into the far cortex to avoid plunging.
Overview[edit | edit source]
Global Impact[edit | edit source]
A systematic review of 204 countries estimated there were 455 million prevalent cases of acute or long-term symptoms of a fracture in 2019.[1] The 2019 global prevalence of fractures of the clavicle, scapula, or humerus was 61 million cases resulting in an estimated 247,000 years lived with disability (YLD). Humeral shaft fractures are the third most common type of long bone fracture and account for approximately 5% of all fractures.[2][3]
A 2015 review paper found that humeral shaft fractures occur in young male adults in 55–63% of cases and a radial nerve palsy is present initially in 10–20% of these patients.[2] A 2020 Nigerian study found that the humerus bone is the predominant location for gunshot and machete cut fractures and that open humeral fractures outnumbered radius and ulnar fractures.[4] The ongoing violence in northern Nigeria may be contributing to the higher number of open humeral shaft fractures observed in this region.[5]
Access to high-quality orthopedic care in LMICs is limited by a lack of providers, resources, and training programs.[6] The global standard is a ratio of one orthopedic surgeon to 200,000 people.[7] In 2008, Uganda reported a ratio of 1 orthopedic surgeon for every 1.3 million people.[8] In 2020, Nigeria had an estimated population of over 206 million people.[9] According to the Nigerian Orthopaedic Association, Nigeria's ratio is 1 orthopedic surgeon to approximately 500,000 people.[7] An estimated 75% of Nigerian orthopedic surgeons are located in 4 major cities which leaves other urban areas and rural regions without coverage.[10]
The national shortages of orthopedic surgeons in LIMCs leaves patients vulnerable to traditional bone setters whose unsafe practices result in worse outcomes compared to no treatment and commonly lead to malunion, limb shortening, gangrene, limb loss, and death.[11][12][13][14][15] A 2007 study of Nigerian healthcare institutions found unacceptably high rates of amputation (57% to 77.8%) and mortality (11.1% to 26.7%) secondary to bone setter's gangrene.[11] A 2004 study on 82 Nigerian patients (median age of 27 years) found that the leading cause for limb amputation (32%) was gangrene resulting from treatment of extremity injuries by traditional bone setters.[12] Since medical officers do not receive adequate exposure to orthopedic surgery during their undergraduate medical education, they often refer fracture patients to traditional bone setters in regions without orthopedic specialist coverage.
In 2022, over 274 million people require humanitarian aid due to emergencies caused by conflict or natural disasters.[16] Nearly 45 million people in conflict zones lack basic access to healthcare.[17] Long bone fractures resulting from penetrating gunshot wounds account for over 25% of injuries sustained in conflict settings.[18] Conflict depletes an already overstretched surgical workforce, as doctors may become internally displaced or are forced to flee.[19] There is a lack of access to high quality surgical education in many conflict zones, as residency training programs either never existed or are closed down due to security risks, economic factors, and a lack of surgical mentors to oversee trainees. Surgeons operating in conflict-affected regions face targeted attacks, often with devastating consequences, which further reduces their numbers.[19][20] The security risks to healthcare workers in conflict zones pose a barrier to recruitment of local and international surgical staff.[19]
A 2017 review paper determined that 87% of injuries requiring treatment after earthquakes were orthopedic injuries, 65% of these injuries were fractures, and 22% were open fractures.[21] External fixator frames have been successfully used for orthopedic damage control for mass casualty events.[22][23] Over a 10 day period after the 2010 Haiti earthquake, orthopedists used external fixator frames to stabilize 73 extremity fractures (48 femoral fractures, 24 tibial/fibular fractures, and 1 humeral fracture) in a field-style operating room where limb alignment was evaluated via manual palpation, intraoperative imaging was not available, and soft tissue care was provided after bone stabilization.
This self-assessed training module uses open source, locally reproducible, high fidelity 3D printed bone simulation models to educate and empower physicians who are not orthopedic specialists to perform modular external fixation procedures as part of the surgical management of open humeral shaft fractures to save limbs and lives. This module teaches essential irrigation and debridement, powered and manual drilling, and modular external fixation skills that are transferable to the performance of other limb-saving and life-saving surgeries that require hardware stabilization and fixation.[24] These skills can be used to prevent needless suffering, disability, and deaths for the estimated 133 million patients who sustain extremity and pelvic fractures globally every year.[1]
Disability Adjusted Life Years (DALY) Burden in LMICs[edit | edit source]
The estimated 2019 DALY burden of the procedure gap for humeral shaft fractures in LMICs is 4.46 million DALY with a 95% uncertainty interval of 3.54 - 5.61 million DALY (Table 1).[1][25][26][27] The estimated YLD portion of the 2019 DALY burden of the procedure gap for external fixation of extremity and pelvic fractures in LMICs is 18.3 million YLD with a 95% uncertainty interval of 12.1 - 26.6 million YLD.[1][26][27] Our DALY and YLD estimates are based on data published in the peer-reviewed literature by authors who are not members of the Tibial Fracture Fixation team. We have outlined our methodology for estimating the DALY and YLD burden in LMICs below:
| YLL | YLD | DALY = YLL + YLD | DALY in LMICs = DALY X 83.8%[26][27] | |
|---|---|---|---|---|
| Estimated Total | 5.30 million | 21,900 | 5.32 million | 4.46 million |
| Lower Limit of 95% Uncertainty Interval | 4.22 million | 13,400 | 4.23 million | 3.54 million |
| Upper Limit of 95% Uncertainty Interval | 6.67 million | 34,200 | 6.70 million | 5.61 million |
A 2021 systematic review of 204 countries estimated there were 156 million cases of new skeletal fractures with a 95% uncertainty interval of 124 - 196 million cases in 2019 (Table 2).[1] A 2021 Swedish Fracture Register study on 295,713 skeletal fractures sustained in adult patients (> 15 years of age) from 2012 to 2018 found 3,267 cases of humeral shaft fractures.[25] Therefore, humeral shaft fractures account for 1.1% of skeletal fractures (3,267/295,713 x 100%) in this national registry. We estimated the annual global incidence of humeral shaft fractures was 1.71 million cases (156 million cases x 1.1%) with a 95% uncertainty interval of 1.36 - 2.15 million cases in 2019.
| Fracture Site | Incidence | 95% Uncertainty Interval |
|---|---|---|
| Fracture of clavicle, scapula, or humerus | 19.3 million | 15.3–24.0 million |
| Fracture of radius or ulna, or both | 30.7 million | 24.5–38.6 million |
| Fracture of hand, wrist, or other distal part of hand | 19.0 million | 15.2–23.5 million |
| Fracture of vertebral column | 8.58 million | 6.64–11.3 million |
| Fracture of pelvis | 6.04 million | 4.44–8.30 million |
| Fracture of hip | 14.2 million | 11.1–18.1 million |
| Fracture of femur, other than femoral neck | 14.6 million | 11.9–18.0 million |
| Fracture of patella, tibia or fibula, or ankle | 32.7 million | 26.9–39.7 million |
| Fracture of foot bones except ankle | 10.7 million | 8.07–14.0 million |
| Total: | 156 million | 124-196 million |
The 2021 Swedish Fracture Register study on 3,267 adult patients with humeral shaft fractures found the 1 year mortality rate was 12.4% for patients ages 63 years +/- 21 years (mean +/- SD).[25] Thus, we estimated years of life lost (YLL) for humeral shaft fractures globally is 5.30 million YLL (1.71 million cases x 12.4% x [88-63 years]) with a 95% uncertainty interval of 4.22 - 6.67 million YLL in 2019 (Table 1).
The 2019 global incidence of fractures of the clavicle, scapula, or humerus was 19.3 million cases with a 95% uncertainty interval of 15.3 - 24.0 million cases resulting in an estimated 247,000 years lived with disability (YLD) with a 95% uncertainty interval of 151,000 – 386,000 YLD.[1] Based on our estimate of the global incidence of humeral shaft fractures (1.71 million cases), we estimate that in 2019, humeral shaft fractures worldwide resulted in an estimated 21,900 YLD (1.71 million cases/19.3 million cases x 247,000 YLD) with a 95% uncertainty interval of 13,400 - 34,200 YLD (Table 1).
The World Bank estimates the total population in LMICs is 6.57 billion which is 83.8% of the world's total population of 7.84 billion.[26][27] Thus, we estimate that the 2019 DALY burden of the procedure gap for humeral shaft fractures in LMICs is 4.46 million DALY (DALY x 83.8%) with a 95% uncertainty interval of 3.54 - 5.61 million DALY (Table 1). Estimates are rounded to three significant figures for counts and one decimal place for percent values.[1]
This module teaches essential irrigation and debridement, powered and manual drilling, and modular external fixation skills that can be used to prevent needless suffering, disability, and deaths for the estimated 133 million patients who sustain extremity and pelvic fractures globally every year.
We estimated the YLD portion of the 2019 DALY burden of the procedure gap for the external fixation of extremity and pelvic fractures globally using previously published YLD estimates of 22.0 million YLD with a 95% uncertainty interval of 14.4 - 31.7 million YLD (Table 3). The World Bank estimates the total population in LMICs is 6.57 billion which is 83.8% of the world's total population of 7.84 billion.[26][27] We estimated the YLD portion of the 2019 DALY burden of the procedure gap for external fixation of extremity and pelvic fractures in LMICs by multiplying the YLD value x 83.8%.
| Fracture Site | Estimated Total YLD | Lower Limit of 95% Uncertainty Interval | Upper Limit of 95% Uncertainty Interval |
|---|---|---|---|
| Fracture of clavicle, scapula, or humerus | 247,000 | 151,000 | 386,000 |
| Fracture of radius or ulna, or both | 210,000 | 131,000 | 325,000 |
| Fracture of hand, wrist, or other distal part of hand | 301,000 | 160,000 | 522,000 |
| Fracture of pelvis | 3.22 million | 2.27 million | 4.43 million |
| Fracture of femur, other than femoral neck | 1.85 million | 1.23 milion | 2.64 million |
| Fracture of patella, tibia or fibula, or ankle | 15.5 million | 10.2 million | 22.6 million |
| Fracture of foot bones except ankle | 480,000 | 291,000 | 750,000 |
| Estimated Total: | 21.8 million | 14.4 million | 31.7 million |
| Total in LMICs (83.8% of Total): | 18.3 million | 12.1 million | 26.6 million |
Open-Source 3D Printing Technologies for High Fidelity Orthopedic Surgical Simulation Training[edit | edit source]
Open-source 3D printing technology supports the local and automated reproduction of the highest fidelity bone simulation models at the lowest cost for medical officers and surgeons who are not orthopedic specialists in LMICs.
Open-source, open filament and user-friendly desktop 3D printers are currently in use at small to medium enterprises, Makerspaces (including but not limited to approximately 2,000 Fab Labs in over 149 countries), start-up incubators, universities, and hospitals worldwide.[28][29][30][31][32][33][34][35][36][37][38][39][40][41][42] Our 3D printed bone simulation models are designed to reduce simulator costs, simplify the simulator build, and minimize simulator assembly time for the learner.
This module provides an open source library of downloadable, 3D printed bone simulation models which accurately represent bone length and diameter, external contour and cross-sectional shape, bicortical anatomy, cortical hardness, cancellous bone porosity, and microstructure, and far cortex thickness for both genders at drilling sites for humeral shaft fractures.[43][44][45][46][47][48][49][50][51][52][53][54][55][56][57][58][59]
All of the module's 3D printed models can be locally reproduced on open source, open filament, user-friendly, fused deposition modelling, single extruder desktop 3D printers that print polylactic acid (PLA), a low-cost, biorenewable, and biodegradable plastic.[60][61][62][63][64] According to one filament manufacturer, 3D printed PLA at 100% infill has a Shore Hardness D value of 83D and 84D while independently measured Shore Hardness D values of 3D printed PLA samples range from 80D to 88D (n=12).[51][52][53] These Shore Hardness D values of 3D printed PLA are within the 3-sigma range for the Shore Hardness D measurements of 86.7D + 1.91D (ave. ± s.d., n=1815) for human cortical bone.[49]
To maximize the likelihood that these 3D printed models provide similar tactile feedback as human bone and do not foster the development of anti-skills, the bone simulation models are made from PLA, a plastic filament with a hardness level similar to cortical bone, which exhibits force and displacement ratios similar to artificial bone, and whose haptic feedback was rated as similar to bone during drilling by experienced maxillofacial surgeons and surgical residents.[49][51][52][53][60][65][66] These bone simulation models are digitally manufactured using customized settings to match age, gender, and bone site-specific cortical thickness values, and cancellous bone porosity values for the target patient population.[2][54][55][56][57][58][59]
We addressed the shortcomings of 3D printed bone simulation models which lack overlying soft tissue and skin simulation layers in teaching the performance of modular external fixation of an open humeral shaft transverse fracture by highlighting the steps that cannot be performed during simulation training but must be performed during the actual clinical procedure in the training objectives, knowledge objectives, and procedure steps of the skills training module page and the checklist of the training logbook module page.
The user's learnings on high fidelity 3D printed bone simulation models will translate into the clinical performance of Modular External Fixation for an Open Humeral Shaft Transverse Fracture which is described in 46 learning objectives on the skills training module page.
Syllabus[edit | edit source]
Pre-Learning Clinical Confidence Assessment[edit | edit source]
Before the learner starts this module:
- Go to this link.
- Download, print out, and complete the pre-learning clinical confidence assessment for this training module.
- Photograph the completed assessment on your cellphone as a backup and file the assessment in your training records.
Phase 1: Knowledge Review[edit | edit source]
It is highly recommended that the learner be familiar with this content before proceeding to the skill pages.
Phase 2: Simulator Build[edit | edit source]
Instructions for 3D Printing Organizations[edit | edit source]
Please pay attention to and follow all the instructions closely to ensure the bone models are printed properly and display the required visual, tactile, and acoustic fidelity for orthopedic surgical simulation training.
- Quick Start Guide for 3D Printing Adult Male Humeral Bone Models
- Detailed Step-by-Step Instructions and Troubleshooting Guide for 3D Printing Adult Male Tibial Bone Models
Simulator Assembly[edit | edit source]
Surgical Equipment[edit | edit source]
Phase 3: Skills Practice[edit | edit source]
Phase 4: Self-Assessment[edit | edit source]
The Humeral Shaft Transverse Fracture Simulator is designed to include mechanisms for targeted feedback which enables the user to: ensure they are practicing the appropriate skills; modify their performance to improve competence; and determine when they have practiced to a sufficient level of mastery to perform the procedure in a patient.
The transparent cellophane that is wrapped around the 3D printed bone models and simulates the overlying periosteum will permit the learner to visually inspect and confirm post-fixation fracture alignment, no Schanz screws perforated the far cortex of the Humeral Shaft Transverse Fracture Simulator, and that the Schanz screws are properly positioned along the humeral diaphysis for the self-assessment framework.
The Training Logbook - Modular External Fixation for an Open Humeral Shaft Transverse Fracture includes:
- Step-by-step procedural checklists to confirm following of the proper modular external fixation technique for open humeral shaft fractures; and
- Visual inspection and taking cellphone photos ("digital X-rays") to verify post-fixation fracture alignment, no pin penetration of the far cortex, and proper pin insertion into the safe zones of the humerus.
Training Module Certificate of Completion[edit | edit source]
Once the self-assessment framework has been completed:
- Go to this link.
- Click on the "Get your certificate" button under the "Training Module Certificate of Completion" section at the bottom of the module page.
- Type in your name, download and print out a certificate of completion for this training module.
- Photograph your certificate on your cellphone as a backup and file the printed certificate in your training records.
Post-Learning Clinical Confidence Assessment[edit | edit source]
After the learner completes this module:
- Go to this link.
- Download, print out, and complete the post-learning clinical confidence assessment for this training module.
- Photograph the completed assessment on your cellphone as a backup and file the assessment in your training records.
If the learner does not feel confident in performing this procedure in real clinical scenarios, the learner can repeat this module and practice the skills training as many times as they feel necessary to gain the confidence to perform this procedure on patients.
Supplemental Learning Topics[edit | edit source]
(Optional) After completion of the module, the learner may wish to learn more about 3D printing technology:
Additional Module Information[edit | edit source]
(Optional) After completion of the module, the learner may wish to learn more about this module:
Follow-on[edit | edit source]
(Optional) After completion and practice to competency, the learner may wish to continue study with these courses:
Acknowledgements[edit | edit source]
This work is funded by a grant from the Intuitive Foundation. Any research, findings, conclusions, or recommendations expressed in this work are those of the author(s), and not of the Intuitive Foundation.
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Global, regional, and national burden of bone fractures in 204 countries and territories, 1990–2019: a systematic analysis from the Global Burden of Disease Study 2019. GBD 2019 Fracture Collaborators. Published:August 20, 2021. DOI:https://doi.org/10.1016/S2666-7568(21)00172-0.
- ↑ 2.0 2.1 2.2 Pidhorz L. Acute and chronic humeral shaft fractures in adults. Orthop Traumatol Surg Res. 2015 Feb;101(1 Suppl):S41-9. doi: 10.1016/j.otsr.2014.07.034. Epub 2015 Jan 17. PMID: 25604002.
- ↑ M. Graves, S.E. Nork. Fractures of the humerus, orthopaedic knowledge update. Trauma, 4 (2010), pp. 201-224.
- ↑ Omoke NI, Ekumankama FO. Incidence and pattern of extremity fractures seen in accident and emergency department of a Nigerian teaching hospital. Niger J Surg 2020;26:28-34.
- ↑ https://www.unocha.org/nigeria/about-ocha-nigeria
- ↑ Conway DJ, Coughlin R, Caldwell A, Shearer D. The Institute for Global Orthopedics and Traumatology: A Model for Academic Collaboration in Orthopedic Surgery. Front Public Health. 2017 Jun 30;5:146. doi: 10.3389/fpubh.2017.00146. PMID: 28713803; PMCID: PMC5491941.
- ↑ 7.0 7.1 Nigeria has 350 orthopaedic surgeons for 170 million citizens. (2016, November 26) Agency Report. Premium Times. Retrieved November 11, 2020, from https://www.premiumtimesng.com/news/more-news/216415-nigeria-350-orthopaedic-surgeons-170-million-citizens.html.
- ↑ Naddumba EK. Musculoskeletal trauma services in Uganda. Clin Orthop Relat Res. 2008 Oct;466(10):2317-22. doi: 10.1007/s11999-008-0369-2. Epub 2008 Jul 16. PMID: 18629599; PMCID: PMC2584282.
- ↑ Population, total - Nigeria [Internet]. Data. The World Bank; 2021 [cited 2021 Dec 10]. Available from: https://data.worldbank.org/indicator/SP.POP.TOTL?locations=NG.
- ↑ National Hospital records high patronage on knee, hip replacement surgeries. (2020, January 26) The Sun Nigeria. Retrieved November 11, 2020 from https://www.sunnewsonline.com/national-hospital-records-high-patronage-on-knee-hip-replacement-surgeries/.
- ↑ 11.0 11.1 Nwadiaro HC. Bone setter's gangrene. Nigerian Journal of Medicine Vol. 16 (1) 2007: pp. 8-10. DOI: https://doi.org/10.4314/njm.v16i1.37273.
- ↑ 12.0 12.1 Umaru RH, Gali BM, Ali N; (2004) Role of inappropriate traditional splintage in limb amputation in Maiduguri, Nigeria. Annals of African Medicine, 3(3):138-140. URL: http://www.bioline.org.br/request?am04034.
- ↑ Dada A, Giwa SO, Yinusa W, Ugbeye M, Gbadegesin S. Complications of treatment of musculoskeletal injuries by bone setters. West Afr J Med. 2009; 28(1):43–7.
- ↑ Mohamed Imad A, Hag EL, Osman Bakri M, Hag EL. Complications in fractures treated by traditional bonesetters in Khartoum, Sudan. Khartoum Med J. 2010;3(1):401–5.
- ↑ Onyemaechi NOC, Onwuasoigwe O, Nwankwo OE, Schuh A, Popoola SO. Complications of musculoskeletal injuries treated by traditional bonesetter in a developing country. Indian J Appl Res. 2014;4(3):313–6.
- ↑ https://gho.unocha.org/
- ↑ Creating Hope in Conflict: A Humanitarian Grand Challenge - Request for Proposals February 19, 2018.
- ↑ Bauhahn G, Veen H, Hoencamp R, Olim N, Tan ECTH. Malunion of Long-Bone Fractures in a Conflict Zone in the Democratic Republic of Congo. World J Surg. 2017 Sep;41(9):2200-2206. doi: 10.1007/s00268-017-4008-5. PMID: 28488041.
- ↑ 19.0 19.1 19.2 Adapted from: https://humanitariangrandchallenge.org/wp-content/uploads/2021/01/Analysis-of-Barriers-Affecting-Innovation-in-Humanitarian-Contexts.pdf
- ↑ International Committee of the Red Cross. Why we can't save her life | On The Frontline. [Internet]. San Bruno (CA): Youtube; 2018 May 3 [cited 2021 Aug 27]. Available from: https://www.youtube.com/watch?v=wm0TYebjyHQ.
- ↑ MacKenzie JS, Banskota B, Sirisreetreerux N, Shafiq B, Hasenboehler EA. A review of the epidemiology and treatment of orthopaedic injuries after earthquakes in developing countries. World J Emerg Surg. 2017 Feb 10;12:9. doi: 10.1186/s13017-017-0115-8. PMID: 28203271; PMCID: PMC5301447.
- ↑ Lebel E, Blumberg N, Gill A, Merin O, Gelfond R, Bar-On E. External fixator frames as interim damage control for limb injuries: experience in the 2010 Haiti earthquake. J Trauma. 2011 Dec;71(6):E128-31. doi: 10.1097/TA.0b013e3182147654. PMID: 21502876.
- ↑ Bar-On E, Lebel E, Kreiss Y, Merin O, Benedict S, Gill A, Lee E, Pirotsky A, Shirov T, Blumberg N. Orthopaedic management in a mega mass casualty situation. The Israel Defence Forces Field Hospital in Haiti following the January 2010 earthquake. Injury. 2011 Oct;42(10):1053-9. doi: 10.1016/j.injury.2011.03.054. Epub 2011 Apr 19. PMID: 21507401.
- ↑ Debas, H. T., P. Donkor, A. Gawande, D. T. Jamison, M. E. Kruk, and C. N. Mock, editors. 2015. Essential Surgery. Disease Control Priorities, third edition, volume 1. Washington, DC: World Bank. doi:10.1596/978-1-4648 -0346-8. License: Creative Commons Attribution CC BY 3.0 IGO.
- ↑ 25.0 25.1 25.2 Camilla Bergh, Michael Möller, Jan Ekelund & Helena Brisby (2021) 30-day and 1-year mortality after skeletal fractures: a register study of 295,713 fractures at different locations, Acta Orthopaedica, 92:6, 739-745, DOI: 10.1080/17453674.2021.1959003
- ↑ 26.0 26.1 26.2 26.3 26.4 https://data.worldbank.org/country/XO
- ↑ 27.0 27.1 27.2 27.3 27.4 https://data.worldbank.org/indicator/SP.POP.TOTL
- ↑ https://world.prusa3d.com/
- ↑ https://3dafrica.org/
- ↑ https://web.archive.org/web/20230912231500/https://impacthub.net/about-us-regions-locations//
- ↑ AIGE Limited. 3D printers. [Internet]. 3D Printers | AIGE Limited. [cited 2021 July 29]. Available from: https://www.aige.info/3d-printers.
- ↑ MoTIV. Motiv tribe [Internet]. MoTIV. 2021 [cited 2021 July 29]. Available from: https://motiv.africa/.
- ↑ https://www.creality3dofficial.com/products/official-creality-ender-3-3d-printer?gclid=EAIaIQobChMIn76Xn4_A9wIVD4FaBR0klwSJEAAYAyAAEgIzRvD_BwE
- ↑ Prusa J. Open-Source 3D printers from Josef Prusa [Internet]. Prusa3D. 2019 [cited 2021 July 29]. Available from: https://www.prusa3d.com/.
- ↑ https://lulzbot.com/store/sidekick747?ref=null
- ↑ Ultimaker. Ultimaker 3D printers: Reliable and easy to use [Internet]. ultimaker.com. [cited 2021 July 29]. Available from: https://ultimaker.com/3d-printers.
- ↑ https://www.fablabs.io/labs/map
- ↑ https://live.fablabs.io/
- ↑ https://wiki.hackerspaces.org/List_of_Hacker_Spaces
- ↑ https://makerspaces.make.co/
- ↑ MoTIV. Motiv tribe [Internet]. MoTIV. 2021 [cited 2021 July 29]. Available from: https://motiv.africa/.
- ↑ Niatech. CoRSU Starts Field Testing 3D Printed Orthopaedic Devices For Children With Disabilities [Internet]. Canada: Niatech; June 13, 2016 [cited 2020 Nov 11]. Available from: https://niatech.org/corsu-starts-field-testing-3d-printed-orthopaedic-devices-for-children-with-disabilities/.
- ↑ Singh, Anudeep & Kumar, Anil. (2014). An Anthropometric Study of the Humerus in Adults. RESEARCH AND REVIEWS: JOURNAL OF MEDICAL AND HEALTH SCIENCES. 3. 76-81.
- ↑ Mall G, Hubig M, Büttner A, Kuznik J, Penning R, Graw M. Sex determination and estimation of stature from the long bones of the arm. Forensic Sci Int. 2001 Mar 1;117(1-2):23-30. doi: 10.1016/s0379-0738(00)00445-x. PMID: 11230943.
- ↑ Akman SD, Karakas P, Bozkir MG. The morphometric measurements of humerus segments. Turk J Med Sci. 2005; 36: 81-5.
- ↑ Papaloucas M, Papaloucas C, Tripolitsioti A, Stergioulas A. The asymmetry in length between right and left humerus in humans. Pak J Biol Sci. 2008 ;11 (21):2509-12.
- ↑ https://3dprint.nih.gov/discover/3DPX-016667
- ↑ https://3dprint.nih.gov/discover/3dpx-016809
- ↑ 49.0 49.1 49.2 Society For Biomaterials 30th Annual Meeting Transactions, page 332. Femoral Cortical Wall Thickness And Hardness Evaluation. K. Calvert, L.A. Kirkpatrick, D.M. Blakemore, T.S. Johnson. Zimmer, Inc., Warsaw, IN.
- ↑ https://3dprint.nih.gov/discover/3dpx-016808
- ↑ 51.0 51.1 51.2 Ultimaker. Ultimaker PLA Technical Data Sheet [Internet]. Ultimaker Support. [cited 2021 July 29]. Available from: https://support.ultimaker.com/hc/en-us/articles/360011962720-UltimakerPLA-TDS.
- ↑ 52.0 52.1 52.2 https://support.ultimaker.com/hc/en-us/articles/360011962720-Ultimaker-PLA-TDS
- ↑ 53.0 53.1 53.2 Vian, Wei Dai and Denton, Nancy L., "Hardness Comparison of Polymer Specimens Produced with Different Processes" (2018). ASEE IL-IN Section Conference. 3. https://docs.lib.purdue.edu/aseeil-insectionconference/2018/tech/3.
- ↑ 54.0 54.1 Forrest AM, Johnson AE, inventors; Pacific Research Laboratories, Inc., assignee. Artificial bones and methods of making same. United States patent 8,210,852 B2. Date issued 2012 Jul 3.
- ↑ 55.0 55.1 National Institutes of Health Osteoporosis and Related Bone Diseases National Resource Center. What is Bone? [Internet]. Bethesda (MD): The National Institutes of Health (NIH); 2018. [Cited 2021 Aug 17]. Available from: https://www.bones.nih.gov/health-info/bone/bone-health/what-is-bone.
- ↑ 56.0 56.1 Meyers, M. A.; Chen, P.-Y. (2014). Biological Materials Science. Cambridge: Cambridge University Press. ISBN 978-1-107-01045-1.
- ↑ 57.0 57.1 Meema HE, Meema S. Measurable roentgenologic changes in some peripheral bones in senile osteoporosis.J Am Geriat Soc 1963;11:1170-82.
- ↑ 58.0 58.1 Bloom RA. A comparative estimation of the combined cortical thickness of various bone sites. Skeletal Radiol. 1980;5(3):167-70. doi: 10.1007/BF00347258. PMID: 7209568.
- ↑ 59.0 59.1 Bloom RA, Laws JW. Humeral cortical thickness as an index of osteoporosis in women. Br J Radiol 1970; 43:522-7.
- ↑ 60.0 60.1 Werz SM, Zeichner SJ, Berg BI, Zeilhofer HF, Thieringer F. 3D printed surgical simulation models as educational tool by maxillofacial surgeons. Eur J Dent Educ. 2018;22(3):e500–5. https://doi.org/10.1111/eje.12332.
- ↑ Legocki AT, Duffy-Peter A, Scott AR. Benefits and Limitations of Entry-Level 3-Dimensional Printing of Maxillofacial Skeletal Models. JAMA Otolaryngol Head Neck Surg. 2017;143(4):389–394. doi:10.1001/jamaoto.2016.3673
- ↑ Liu, K., Madbouly, S. A., Schrader, J. A., Kessler, M. R., Grewell, D. and Graves, W. R. (2015), Biorenewable polymer composites from tall oil-based polyamide and lignincellulose fiber. J. Appl. Polym. Sci., 132, 42592. doi: 10.1002/app.42592.
- ↑ Mills, C. A.; Navarro, M.; Engel, E.; Martinez, E.; Ginebra, M. P.; Planell, J.; Errachid, A.; Samitier, J. J. Biomed. Mater. Res. A 2006, 76A, 781.
- ↑ Auras, R.; Harte, B.; Selke, S. Macromol. Biosci. 2004, 4, 835.
- ↑ Husemoglu R. B., Baysan G., Ertugruluoglu P., Tuc Yucel A., Havitcioglu H. , The Mechanical Comparison of Artificial Bone and 3D Printed Bone Segments, Journal of Medical Innovation and Technology, 2020; 2 (2):127-130.
- ↑ Haffner M, Quinn A, Hsieh TY, Strong EB, Steele T. Optimization of 3D Print Material for the Recreation of Patient-Specific Temporal Bone Models. Ann Otol Rhinol Laryngol. 2018 May;127(5):338-343. doi: 10.1177/0003489418764987. PMID: 29667491.