
An ongoing project designed and implemented at Universidad Tecnológica de Coahuila in Parras de la Fuente, Coahuila México to provide safe drinking water purified by the process of solar distillation.
Numerous different methods exist for purifying water, all with the same intended result: to obtain water that is safe for humans to drink, referred to as potable. Some of the most common methods of water purification are boiling, addition of chemicals (iodine or chlorine), reverse osmosis, and filtration. All of these methods are effective, but may require resources that are unavailable in some locations. For example, while it may be very difficult to obtain chemicals or maintain a fire in remote areas, sunlight is often a readily available resource usable for water distillation.
This document is a proposal to implement a passive solar distillation system prototype as a working example of utilizing energy from the sun to provide safe drinking water for human consumption.
Theory[edit | edit source]
Passive solar water distillation is considered an appropriate technology because it operates on the same principles that produce rainfall, purifying water through a process of evaporation, condensation, and collection. Solar distillation could be considered a form of bio-mimicry because of its relation to the rain cycle and the greenhouse effect. The solar distillation process can be performed on brackish, salty water or from sources such as rain, municipal, well, or spring. By utilizing the plentiful energy of the sun to evaporate water, dissolved metals and minerals, such as arsenic, barium, cadmium, chromium, and lead are separated from contaminated water [1]. Distillation also removes salts and biological contaminants such as E.Coli[2].
A conventional solar distiller is a box with a glass roof, referred to as a glaze, set at and angle from the horizontal to ensure optimal sun exposure. This angle is roughly equal to the latitude of the location. The distiller faces south if in the northern hemisphere, north in the southern hemisphere[3]. Untreated water is routed into a holding basin inside the distiller. Radiation from sunlight penetrates the glass and heats the inside of the distiller, causing the water in the basin to evaporate. The absorption of higher frequency radiation heats the water and solar still internals. The glaze traps infrared re-radiation causing a greenhouse effect resulting in higher temperatures [4]. The evaporation process separates contaminants from the water and results in a thin condensate on the underside of the glass cover. The condensed, distilled water then runs off the glass into a trough, and is transferred to a water storage container for domestic use. Contaminants and particulates remain in the basin and must be washed away periodically.
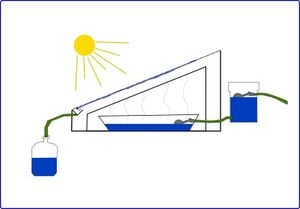
Construction plans[edit | edit source]
Scott Harris and Jeffrey M. Hinton, participants of the Parras 2005 program constructed a solar water distiller and provided a final analysis of their project on 5 August 2005. The unit they constructed is located in the laboratory at the Universidad Tecnológica de Coahuila, Parras Campus. This project provides a solid basis for further research and development of small scale water distillation in Parras. However, after reviewing their final analysis, construction infrastructure, and materials used, we propose some design changes and plan to research different building materials. Modifications include the substitution of the insulation material, floor coating, basin structure, and design of the collection trough. The intention is to improve upon the existing design, utilize more affordable, durable materials, and research additional methods for increasing efficiency. Portability is not a consideration with this prototype design. The materials and design chosen do not lend themselves to easy transport. The glaze is fragile, the wood veneer internal frame is frail, and the metal basin is not secured. This unit will only operate at the UTC campus, subsequent iterations of this design should consider portability issues.
| Function | Current material | Possible alternatives | Justification | Possible issues |
|---|---|---|---|---|
| Insulation | Styrofoam | Lechugilla fiber, Wool, Clay/straw mixture [5], recycled denim, Perlite [Details], Fiberglass as a last resort. | A natural insulator would be preferable, and the Styrofoam tends to melts with heat | Flammability, water absorption, R-Value, cost, availability, lifespan |
| Collection Trough | PVC pipe | metal, ceramic, wood | The PVC melts from the heat | Heat resistance, Conductivity removing heat from the system |
| Black coating for heat retention | Spray paint | Ceramic tile, black woven tarp, tinted glass | Spray paint may be the only solution, but a more natural black coating would be preferable | Chemical leeching, heat resistance, fragility |
| Untreated water basin | Spray painted galvanized metal | Stainless steel, Ceramic | Spray paint and silicone caulking may contaminate water distillation process | Difficulty obtaining high temperature black sealant. Finding basin of proper size |
| Internal frame | Thin wood veneer | Thicker wood with sturdier construction, or metal | Veneer is too thin, lacks structural support | Affordability, availability, weight, ease of color adhesion |
Proposed location[edit | edit source]
This passive solar water distiller experiment is intended for Universidad Tecnològica de Coahuila campus located on Madero Street, Parras, Coahuila. UTC has a student, faculty, and employee population of 205 people, comprising a continuous demand for potable water. The water demand is important because it directly affects the design of the distiller in respect to desired quantity of water output.
The public supply is the most likely source of water. Although municipal water has been given some treatment, it is not always potable. Municipal water is currently the easiest method of water acquisition. In the future a rainwater catchment system could be implemented. At this time, the location of the distiller on the UTC campus is undetermined pending investigation of the best possible placement. A flat and level area with direct access to water and sun is necessary. considerations must be taken to ensure it is not in a place where the glaze can be easily damaged by vandalism or accidental breakage. The system must be located in an area that facilitates easy access and maintenance.
Justification[edit | edit source]
Currently, the UTC purchases their drinking water from SierrAzul in 5-gallon jugs (19-litres) at a cost of approximately $1.40 USD ($14.50 pesos) each. Potential benefits of implementing this system are reduced cost of water and self-sufficiency. Assume the cost of the system is $106.92 + $79.32 USD = $186.24 USD. This is the cost of materials from the previous year added to the total cost from the second iteration. If UTC consumes one 5-gallon jug of water per day, the system would pay for itself in approximately 133 days based on the savings from not purchasing the water. The presence of this project at UTC may influence students to pursue additional education in solar technology, possibly taking the knowledge with them in their daily lives, and into the future.
If we successfully implement the solar water distiller at UTC, there may be increased potential for expansion and installation to other locations in Parras. Solar water distillation may apply to small systems for homeowner usage, as well as larger installations in businesses or agricultural locations.
Project update: week 5[edit | edit source]
Physical progress[edit | edit source]
Week 4: Disassembly initiated. Melted components partially removed and inspected. These components include Styrofoam, PVC pipe, and plastic coating.
-
Fig 1: Distiller with glaze removed, no modification.
-
Fig 2: Much of the styrofoam is shrunken and hardened from excessive heat.
-
Fig 3: Sawdust may be a decent insulator but it's very messy when uncontrolled. Also encountered were styrofoam peanuts, and various loose ceramic tiles. *Materials
-
Fig 4: Silicone glue was used extensively. *Materials
-
Fig 5: PVC did not survive the heat. *Materials
-
Fig 6: Melted collection trough, with glue.
-
Fig 7: A closeup of insulation surrounding the untreated water basin.
-
Fig 8: Insulation underneath the basin was also melted.
-
Fig 9: Wooden internal frame that holds water basin. Constructed from thin veneer.
-
Fig 10: After breaking up and discarding the large pieces of styrofoam, the smaller pieces must be disposed of.
Proposed materials[edit | edit source]
Insulation[edit | edit source]
Insulation is by far the most difficult material to choose. Many options are available, and we have not yet made a decision what material will be most appropriate when comparing cost, availability, and effectiveness. Some possible solutions:
Notes:
- Any flammable insulator may require additional measures to prevent fire.
- Fire protection means some type of sealing and/or chemical treatment of loose-fill insulation.
- Insulation should have minimal or no contact with both liquid and vaporized water.
- A fairly inexpensive fire retardant and insect deterrent is
- 2.5 ounces borax mixed with 2 cups boiling water [6]
| Material | Justification | Concerns | Estimated R Value [7] |
|---|---|---|---|
| Cotton fiber from denim factory | Locally available | Possible toxic chemicals (dyes, etc), fire hazard. | 3 to 3.8 (Cellulose) |
| Perlite - Loose fill [8] [9] | Common material, naturally occurring glass, hopefully available from [Parras Farm Stores]. High temperature tolerance. | price, availability, possible water contamination | 2.7 |
| Vermiculite [10] | Very similar to Perlite, fire resistant | May contain asbestos [11] | 2.13 to 2.4 |
It may be useful to combine a solid insulator with some type of loose-fill [12] or spray-in-place [13]
Water basin[edit | edit source]
The water basin is fairly small, spray painted black, and sealed with silicone glue. Some ideas for improvement:
- Fabricate or purchasing a larger basin
- Search for possible alternatives to metal basin
- Reuse a dark colored sink basin
- Fabricate a ceramic basin
- Line current basin with tinted glass or dark ceramic tile
- :The additional weight is acceptable for this prototype distiller due to the stationary design. Additionally, the water basin can be removed if transportation is necessary.
- Search for possible alternatives to metal basin
- Purchase a second identical basin and place the two side by side
- Remove spray paint
- Find an alternative non-toxic black paint, with high temperature tolerance.
- If a metal trough is the only choice, research other darkening solutions such as patination, or staining.
- Look into the possibility of using a wick system to draw untreated water into the still.
Collection trough[edit | edit source]
This could be replaced with fabricated metal, ceramic, or another material with high temperature durability. Considerations should be taken regarding the conductivity of the material chosen.
Fasteners and sealant[edit | edit source]
Much of the exiting glue needs to be removed and replaced with sturdier fasteners such as brackets and screws. The chosen material available in this region is high temperature RTV silicone gasket sealant.
Project update: week 8[edit | edit source]
Week 6[edit | edit source]
- Implemented plumbing for water intake system including external storage bucket and basin float valve system.
- Determined insulation to consist of perlite. Visited several local nurseries in town and found a nursery that can order bulk perlite.
- Visited local construction stores for black high temperature silicone. Not available locally.
- Instead purchased two small tubes of red high temperature caulking from Home Depot in Torreón.
Week 7[edit | edit source]
- Finalized materials list
- Went to Home Depot in Torreón to purchase materials
- Proposed budget-to-date for distiller is $100.00 (US)
- Money spent to date is $35.00 (US)
- Ordered Perlite from nursery. Two large buckets are currently on order and should arrive Thursday 10/13. Because of a planned trip, they must be picked up Saturday 10/15.
Automatic water intake system[edit | edit source]
- Implemented Sunday, 9 July 2006
-
Fig 1: Float valve inside the external holding bucket
-
Fig 2: Holding bucket outtake
-
Fig 3: Drilling the intake hole for the water basin
-
Fig 4: Installing the water basin float valve
-
Fig 5: Testing float valve operation
-
Fig 6: Siphoning water from the basin to open the float valve
-
Fig 7: Water basin in action
Project update: week 10[edit | edit source]
Summary of purchased items[edit | edit source]
| Descripción | Description | Qty | Cost (Pesos) | Total |
|---|---|---|---|---|
| Cubeta Multiusos | 5 Gal Bucket | 1 | $69.00 | $69.00 |
| Silicon Rojo | Red Gasket Silicon | 2 | $28.50 | $57.00 |
| Valvula 3/4" | 3/4" Valve | 2 | $38.50 | $77.00 |
| Union Por Manguera | Hose union | 3 | $25.00 | $75.00 |
| Flotador #3 Por Tanque | Toilet float valve | 2 | $17.91 | $35.82 |
| Spray Master Paint | Spray Master Paint | 1 | $19.00 | $19.00 |
| Tapa Por Cubeta | Bucket Top | 1 | $25.00 | $25.00 |
| Manguera Jardin 1/2" 3m | 1/2" Garden Hose 3m | 1 | $16.80 | $16.80 |
| 1 Hoja Triplay 9mm | One Sheet 3-Plywood 9mm | 1 | $290 | $290 |
| 1 Hoja Triplay 60cm x 1.80 cm | One Sheet of 9mm 3-Plywood | 1 | $109 | $109 |
| Barrotitos 1x2x1.46 | Misc 1"x2" | 2 | $10 | $20 |
| Pijas 1/2" | Screws | 80 | $.28 | $28 |
| Pijas varios | various screws | 50 | $.35 | $17.30 |
| Cinta Medir 3m | 3m Measuring Tape | 1 | $30 | $30 |
| Collecionador de agua metale - hecho de encargo | Metal water collector - custom built | 1 | $95 | $95 |
| Total | $935.92 | |||
| Estimated US Dollars (11.8:1 Pesos:Dollar) | $79.32 | |||
Construction[edit | edit source]
-
Cutting wood with the help of Mari Angél
-
Measuring internal frame dimensions
-
Securing the internal frame
-
More internal frame construction
-
Adding black plastic used for heat retention
-
Trimming screws with a grinder
-
Measuring dimensions of the insulation cover
-
Cutting plastic for the insulation cover
-
Angela about to cut wood
-
Securing the original plastic (removed later)
-
Ready to present but not ready to use
-
Replacing the plastic
-
Complete, minus the glaze
-
Almost ready to test!
Testing[edit | edit source]
Day 1[edit | edit source]
- Cloudy in the morning. Rain storm later in the day.
- The entire distiller got wet and the underside is saturated.
- No water was evaporated.
Day 2[edit | edit source]
- Finished setup in the morning.
- Moved the distiller to face south and maximize sun exposure. Because of this, the system is no longer in reach of the nearest spigot and the storage bucket had to be filled manually.
- The glaze was not properly sealed until 5:00 pm.
- Water constantly escaped and no evaporation occurred.
- After the glaze was sealed, evaporation began immediately.
- About 10ml of water was successfully distilled and recovered.
- A significant amount of water was lost because the plastic tube used to catch water drops and deposit them in the collector was not completely sealed to the glass. Additionally, the circular shape of the tube may be allowing for water droplets to slip past and fall on the wood frame at the bottom of the glaze.
- Due to other commitments such as class and lunch, temperature readings were sporadic. All temperature data had to be thrown out because the system was unsealed and not retaining heat.
Day 3[edit | edit source]
- Sealed the bottom of the water basin to prevent leakage
- The basin is not properly sealed in the lower corners and continues to leak.
- Sealed hose connection to the basin using Teflon tape.
- Added more gasket seal to the rubber hose used to drip water into the trough.
- Cut the hose in half to decrease the chance of droplets falling past the collection trough.
- Successfully increased the probability of distilled water falling into the trough.
- Completely sealed the glaze using high temperature silicon gasket seal and duct tape.
- Metal tape would be a better choice, but after 3 unsuccessful hours of searching Parras, duct tape was the only choice.
- Distilled a very small amount of water in the remaining daylight hours.
Day 4[edit | edit source]
- Full day of sunlight with some cloud cover
- By the end of the day, approximately 500ml of water was distilled.
- Appearance of distilled water: water is clear, and smells like plastic.
- Taste test: Jeff Kinzer and Eddie Durán were the subjects. The consensus is that the water tastes terrible, like plastic with a cotton aftertaste.
- Problems encountered:
- The untreated water basin continued to leak, causing separation and warping of the wood in the front bottom area of the distiller.
- While removing the glaze to move the unit into storage, one corner of the glass broke.
Instructions[edit | edit source]
Use[edit | edit source]
- Connect hose from water source to bucket. Alternatively, connect the hose directly to the system. The bucket is solely to provide temporary storage if the water source must be disconnected and used for another purpose.
- Turn on water and monitor basin and bucket as they fill
- Wait patiently
- Change clean water receptacle when full
Maintenance[edit | edit source]
- Wash untreated water basin periodically
- Clean glaze
- Check for hose leaks
- Cover with tarp if it rains
- Replace float valves as necessary
- Check glaze seals
- Check hose connections for leaks, repair if necessary
Possible points of failure[edit | edit source]
- Water collecting in the bottom of the system
- Degradation of the glaze seal, which will result in loss of efficiency and increase the possibility of contamination from outside sources.
- Hose connection leakage
- Degradation of the wood frame from water leakage and exposure to the elements
Future changes and upgrades[edit | edit source]
- Fix thread mismatch - The float valves use a fine thread, but hose connectors use coarse thread. This mismatch causes unreliability of connections, and constant leakage. Currently several windings of Teflon tape are in place to stop the leakage. The best solution is a thread adapter or purchasing fine thread hose connectors.
- Construct the system out of something other than wood, especially not particle board - Metal or cement may hold up better than wood when exposed to rain, wind, and high amounts of sun.
- Use a different method of depositing condensed water droplets in the collection trough. The tube is not completely effective - One possible solution is a curved piece of plastic running from the bottom edge of the glaze diagonally into the collection trough.
- Install easy hose connect/disconnect in the rear of distiller - The system should be easy for anyone to connect or disconnect.
- Completely seal perlite insulation from the water chamber - Currently silica powder from the crushed perlite is contaminating the entire water evaporation chamber.
Conclusion[edit | edit source]
What succeeded[edit | edit source]
- Replacement of the internal frame
- the addition of black plastic reused from defective water storage tanks, as a replacement for spray paint
- Float valve water intake system
- Perlite insulation as a replacement for styrofoam
- Having access to tools, help and transportation thanks to Angela's host, the Madero family. Without them this project would not have been possible to implement.
What failed[edit | edit source]
- water input systems
- bucket with float valve is unnecessary
- However, the extraneous bucket has been essential for testing because we do not have a long enough hose to reach from the nearest spigot to the testing site.
- The extra materials also provide a replacement float valve and extra hose parts for future maintenance.
- Thread mismatch with hose unions -- fine thread on float valve, coarse thread on the hose connector.
- hose coupling leaks (currently using Teflon tape and drip catch)
- The water basin leaks, attempts to seal it were unsuccessful.
- Insulation is not completely sealed. Perlite enters the distillation chamber and silica particles settle in the untreated water basin.
- The glaze is very difficult to seal because the box frame is warped and is continuing to deform.
- Attempts to seal the glaze with high temperature silicone gasket seal were partially successful.
- More silicone is required to seal the unit than we were able to purchase.
- Metal tape would suffice for sealing the glaze. Finding the tape in Parras is difficult, and we were not able to encounter any. More research should be done regarding the safety of using aluminum tape in possible contact with the distilled water.
Final notes[edit | edit source]
As a functional unit, this project was only partially successful. There are many issues that still need to be solved regarding the efficiency, ease of use, and maintainability of the distillation system. The water recovered from the distiller has not been properly tested, but this water is not likely to be much safer for consumption than the original untreated water.
As a continuation of the work from the previous year, the project has progressed. Several components, such as the water intake system, internal frame, insulation, and collection trough, have improved greatly. The addition of a float valve to allow for automatic water input solved one of the major problems encountered originally. The distiller no longer must be filled manually, and there is a much lower chance of the system going dry and melting internal components. The internal frame is now sturdier, and is using black plastic rather than spray paint to maintain internal temperature. The perlite insulation is far more resistant to heat than styrofoam. The collection trough is fabricated from metal and will not melt as did the previous PVC trough.
This project has good prospects of improving significantly in the years to come. The next group to work with the solar distiller will have two years of experience to draw from in designing and implementing the system which may one day provide safe drinking water for Universidad Technológia Coahuila in Parras de la Fuente.
References[edit | edit source]
Solar distillation
- Wikipedia:Solar distillation
- http://www.solaqua.com/solstilbas.html#cap
- https://web.archive.org/web/20061010224651/http://www.wot.utwente.nl/documents/publications/1990_ssadc/ssadc/chapter3.htm
- Solar Distillation in Parras 2005 (not available online)
Distillation background
- https://web.archive.org/web/20120915003611/http://www.nsf.org:80/consumer/drinking_water/selecting_dwtu.asp?program=WaterTre
- https://en.wikipedia.org/wiki/Greenhouse_Effect http://en.wikipedia.org/wiki/Greenhouse_Effect
- http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/grnhse.html#c1
Perlite
- http://www.perlite.info
- https://web.archive.org/web/20121127174713/http://www.perlite.org:80/perlite_info.htm
- Wikipedia:Perlite
Vermiculite
Asbestos
Insulation






























