The Design and Development of a Solar Powered Refrigerator
R.H.B. Exell, Sommai Kornsakoo, D.G.D.C Wijeratna. Bangkok, Thailand: Asian Institute of Technology, 1976.
This book was ported by Jianlang Mai, Scott Gennings, Graham Coote, Ana Lise Herrera and Howard Swartz.
Preface
This research report describes work on the development of a solar powered refrigeration system which will eventually lead to the production of a village size ice maker or to a cold storage unit for food preservation.
This subject was examined by Mr. D.G.D.C. Wijeratna in his Individual Studies Project Report (No. 34), and the experimental unit was designed by Dr. R.H.B. Exell. The construction and testing of the unit was by Mr. Sommai Kornsakoo for his Master Degree Thesis.
The Asian Institute of Technology (AIT) is indebted to the John F. Kennedy Foundation, Thailand, for financial support in the form of a grant for solar energy research made in response to a proposal made in 1973 by Professor H.E. Hoelscher, President of AIT, to Dr. Tbanat Khoman, Chairman of the Foundation.
Summary
A small ammonia-water intermittent absorption refrigerator with a 1.44 m2 flat plate solar collector has been tested as a first step towards the development of a village ice maker. No oil or electricity is used. Regeneration takes place during the day and refrigeration at night. Rapid absorption is obtained by means of a new feature, first proposed by Swartman, in which the heat of absorption is dissipated from the flat plate.
In the generator 15 kg of solution containing 46% ammonia in water are used. On a clear day the solution temperature rises from 30oC, to 88oC and 0.9 kg of pure ammonia is condensed at 32oC. During refrigeration the temperature of the ammonia drops to -7oC. The estimated overall solar coefficient of performance (cooling effect divided by solar heat absorbed) is 0.09, which though small is comparable with previously published work. Developments in the design are discussed.
Contents
| Preface | ii |
| Summary | iii |
| Contents | iv |
| I. INTRODUCTION | 1 |
|---|---|
| The Basis for Considering Solar Energy | 1 |
| Objectives of the Study | 1 |
| Possibilities for Research and Development | 2 |
| The Rationale for Selecting Solar Refrigeration | 4 |
| II SOLAR REFRIGERATION | 8 |
| Indices of Performance | 8 |
| Operation of the Intermittent Ammonia-Water System | 9 |
| Analysis of the Ideal Cycle | 9 |
| Rigorous Analysis of the Ammonia-Water Cycle | 13 |
| Historical Development | 15 |
| III DESIGN OF THE EXPERIMENTAL UNIT | 23 |
| Choice of Configuration | 23 |
| Operation of the System | 24 |
| Concentration of Aqua-Ammonia | 26 |
| Regeneration Phase of the Cycle | 26 |
| Refrigeration Phase of the Cycle | 28 |
| Collector-Generator Specifications | 28 |
| The Volume of the Generator | 30 |
| The Size of the Receiver for Ammonia | 32 |
| Heat of Generation | 32 |
| Heat of Condensation | 33 |
| Further Details of the Design | 33 |
| IV EXPERIMENTAL TESTS | 37 |
| Relationship between Plate Temperature and Solution | |
| Temperature | 37 |
| Experimental Results | 37 |
| Amount of Ammonia Distilled | 52 |
| Cooling Ratio 54 | |
| Heat Absorbed by Solution During Regeneration | 55 |
| Solar Coefficient of Performance | 56 |
| Discussion | 56 |
| V CONCLUSIONS AND PLANS FOR CONTINUING RESEARCH | 59 |
| Conclusions | 59 |
| Economic Considerations | 59 |
| Modifications | 60 |
| The Development of a Village Ice-Maker | 60 |
| Alternatives | |
| References | 65 |
| Appendix A | |
| Charging - Equipment - Procedure | 67 |
| Appendix B | |
| Estimation of Incident Solar Radiation | 72 |
I INTRODUCTION
The Basis for Considering Solar Energy
There are several important reasons for considering solar energy as an energy resource to meet the needs of developing countries. First, most the countries called developing are in or adjacent to the tropics and have good solar radiation available. Secondly, energy is a critical need of these countries but they do not have widely distributed, readily available supplies of conventional energy resources. Thirdly, most of the developing countries are characterised by arid climates, dispersed and inaccessible populations and a lack of investment capital and are thus faced with practically insuperable obstacles to the provision of energy by conventional means, for example, by electrification. In contrast to this solar energy is readily available and is already distributed to the potential users. Fourthly, because of the diffuse nature of solar energy the developments all over the world have been in smaller units which fits well into the pattern of rural economics.
Objectives of the Study
The present study is part of a project in solar energy utilization in AIT, aimed at the development of one or more prototype units demonstrating the usefulness and economic viability of solar energy for the designed purposes.
The specific objective of the argument in this chapter is to identify an area of solar energy utilization useful to the developing countries of Asia, and further, to select a suitable device for development and for a preliminary investigation.
Possibilities for Research and Development
Solar energy research seems to have gathered momentum during the last two decades. Over this period there have been many publications, seminars and conferences dealing with solar energy. One of the most up to date and comprehensive surveys of solar energy applications is a report by an ad-hoc advisory panel of the Board on Science and Technology for International Development entitled 'Solar Energy for Developing Countries: Perspectives and Prospects', NATIONAL ACADEMY OF SCIENCES (1972). The conclusions of this report supersede those of earlier such surveys and are summarized below.
The panel observes that solar evaporation has been a historical, traditional method of obtaining salt from sea water or brines; it remains important today on both a small and large scale in many countries. There appears to be little research that cannot as well be done by the industries using this process.
Water heating technology is well established and the needed development is largely to adapt the technology to use materials and manufacturing capabilities of the country in question. Hot water for hospitals, schools and other such institutions and families could become much more widely available with these developments. The nature of the equipment is such that it can be manufactured in developing countries, and adapting it to their conditions seems straightforward.
Solar distillation must still be regarded as experimental but small scale community stills are near to extensive commercial applications. Designs are now available for solar stills that are serviceable and can be used with a reasonable degree of confidence. Further research in this application would involve adaptation of existing technology to the specific needs of developing countries through design modifications to allow the use of locally available materials and locally manufactured components.
A traditional and widespread use of solar energy is for drying, particularly, of agricultural products. The design and control of these for particular crops or other materials to be dried are areas of research that could lead to more practical applications in developing countries which could result in improved utilization of food supplies.
Research and development in solar heating have been aimed almost entirely at applications in the temperate climates of industrialized countries. The panel knows little of the real extent of needs for space heating in developing countries, or of the possible role of solar energy in meeting these needs.
Studies in air conditioning aimed primarily at United States and Australian applications are still in early stages. Technological feasibility appears to be assured; economic feasibility is now under study. The best methods of obtaining cooling with solar energy in developing countries are far from clear at this time and the immediacy and extent of needs for air conditioning are not known.
There are many refrigeration cycles and systems that can be considered for solar refrigeration. It has yet to be established what may be the best scale on which to operate solar refrigerators in developing countries. There are a substantial number of open questions regarding refrigeration, and the application has the attractive possibility of better utilization of available foodstuffs if refrigeration could be successfully provided.
The possible applications of the successful development of economic solar energy conversion to meet needs for mechanical or electrical energy are wide. This conversion remains an elusive yet intriguing problem. Solar cooking appears to be simple in its technology and significant in its advantages if it can be successfully applied, Solar cookers have been developed to a degree of satisfactory technical performance for providing at least part of the cooking needs of families. However, extensive field trials in India, Mexico and Morocco have so far not resulted in social acceptance of these devices. The conclusions at the Panel are summarised thus: the solar processes that are now useful or that could be brought to a stage of development in which they could produce useful results in the shortest time are evaporation, drying, distillation and water heating. More extensive development in refrigeration, solar heating, cooling and thermal design of buildings should make some of these uses practical within the decade. Applications of solar power will require the substantial development of new technology.
The Rationale for Selecting Solar Refrigeration
The discussion in the earlier section indicates that out of the solar energy applications that are in the experimental stage, space heating and air conditioning are not high priority needs in Asia, conversion to mechanical or electrical energy is best left to be carried out by well-equipped laboratories of industrialised countries and that the social acceptance of solar cooking is rather doubtful, This leaves solar refrigeration as a possible area of study. In order to select a device for further development the following questions must be answered. What is the need for such a device in developing countries, especially, in Asia? Should the device be a food cooler or an ice maker? What size should it be? The rest of this section is devoted to an attempt to find answers to these questions.
A distinguished solar scientist of South East Asia writing anonymously makes the following comments in an article entitled 'A Case for a Solar Ice Maker', ANON., (1963).
"After eight years of study of the problems of applying solar energy in an underdeveloped country I believe that the most promising line of research is to develop an ice making machine. The goal should be a self contained, reliable ice making machine capable of making at least 10 lbs. of ice per sunny day at a cost of one U.S. cent per pound using only solar energy and water as inputs. In tropical countries vast amounts of fresh fruit, vegetabies and fish are lost or their value depreciated by spoilage. This spoilage could be prevented by freezing them with ice... Ice is an important commodity of commerce, fetching as much as 10 U.S. cents per pound in remote areas because of its high cost of transportation (due to melting en-route or the alternative high cost of making it locally at the remote place by electricity or fuel). A foreign made electric refrigerator costs about 250 $ U.S., the cost of a comparable solar icemaker would be at least 250 $ U.S.It may seem strange that a solar ice maker costing 250 $ U.S. would be bought when people were not buying solar cookers at only 10 $ U.S. each. The explanation is that the solar ice maker would be bought by traders and shop owners who can easily afford the amount and they would use the ice for preserving their valuable stocks of fresh fruits, fish etc...... Also the poor people who produce the fresh fruit, fish etc., can afford to buy ice at about one or two U.S. cents per pound, as it is only a small short-term investment of about 10 or 20 U.S. cents, which they can recover within a few days after the sale of their frozen products".
BA HLI et al. (1970) have studied the possibilities for the development of ice makers in Burma. They state that solar ice makers and refrigerators are assured of success purely from the meteorological point of view. They also observe that the solar ice making facilities can either be of domestic size or of community size for local conditions. The domestic solar ice makers and refrigerators must be as automatic as possible in order to compete with electrically operated refrigerators. Community size solar ice makers can have manual participation because there would be an operator available for each ice maker. Roughly, the cost of production of ice by local factories is half U.S. cent per pound of ice ex-factory, but the cost of ice in the hand of the consumer would be about one U.S. cent per pound in the cold season and much higher in the hot season. They conclude that under these conditions, if a solar ice maker can make ice anywhere in Burma for about one U.S. cent per pound that ice could be a boon to the country.
MERRIAM (1972) discussing possible applications of solar energy in developing countries observes thus:
"A very promising application is refrigeration. Refrigeration encompasses household refrigerators, space cooling, air conditioning of buildings etc., but I have chosen to concentrate attention on one particular possible device, a machine for making ice. This is for several reasons, both technological and socio-economic. For one thing, converting the solar radiation into ice solves the problems of intermittency and storage. Ice can be stored for months. Also it is transportable,... An ammonia-water cycle is contemplated..... Several ice makers and refrigerators using this cycle and solar energy input have been built, The design I have in mind would be constructed of mild steel, and would be rugged and simple without moving parts. The output would be 60-70 kg/day of -10oC ice, the input would be l0-12 m of solar radiation and the services of a full-time unskilled operator".
The answers to the questions raised at the beginning of this section can now be provided, viz:
Solar refrigeration is one of the most promising fields for further development; An ice maker seems to be the most useful device in developing countries; If ice can be made at about one U.S. cent per pound commercial viability is assured. A community size unit producing loo-150 lbs. of ice a day, with some manual operation is to be preferred for initial development; domestic refrigerators need to be automatic as far as possible.
The first objective of the study has been reached with the above conclusions, i.e., that of selecting a suitable device for further development. The next step is to make a preliminary study aimed at the development of a community size solar ice maker. As a first step towards this goal an experimental ice maker will be designed and built which will provide experience for further development.
II SOLAR REFRIGERATION
Some of the theoretical concepts that are useful in analysing the performance of solar refrigerators will be presented in this chapter. A brief analysis of the ammonia-water cycle will be made as this will be used in the experimental refrigerator.
Indices of Performance
Any solar cooling device essentially consists of two parts: a cooling unit employing a thermodynamic cycle no different from that employed in conventional refrigerators, and a solar heat source with a flat-plate or a focusing collector to operate it. The usual index by which the performance of a refrigerator is measured is the coefficient of performance which is defined as the ratio of cooling produced to heat supplied. This same concept may be applied to the refrigerator component and a cooling ratio may be defined as
The performance of the solar collector can be defined by a heating ratio given by
The overall performance ratio can now be defined as the product of the two above defined ratios, or explicitly as
The concepts of heating ratio and cooling ratio are especially useful when analyzing systems where the collector and generator are separate.
Operation of the Intermittent Ammonia-Water System
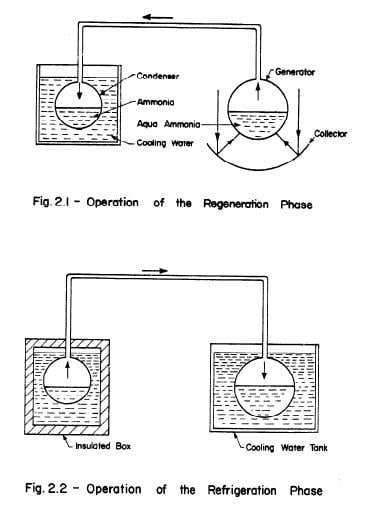
Figures 2.1 and 2.2 show a simple system consisting of two vessels connected by an overhead pipe. The right hand vessel contains aqua-ammonia and functions as the generator-absorber. The left hand vessel contains pure ammonia and functions as the condenser-evaporator.
The operation of the intermittent aqua-ammonia system can be divided into two phases: the regeneration phase and the refrigeration phase. During the regeneration phase heat is supplied to the generator-absorber which contains an ammonia solution of high concentration. As the solution is heated the pressure rises and once condensation pressure is reached ammonia distills off and condenses in the evaporator-condenser which is immersed in a bucket of water, Fig. 2.1.
During the refrigeration phase the heat source is removed and the generator-absorber is allowed to cool. The pressure drops and the ammonia starts evaporating drawing heat from the surroundings thus producing cooling. The weak ammonia solution in the generator-absorber absorbs the evaporated ammonia and the process continues until all the ammonia in the condenser is evaporated, Fig. 2.2.
Analysis of the Ideal Cycle
In the following analysis of the ammonia-water absorption cycle all thermodynamic processes are assumed to be reversible.
Energy is transferred in the form of heat at three temperature levels, i.e.,
- atmospheric temperature Ta, at which heat is rejected in the condenser and absorber,
- the temperature at which heat is taken from the cold chamber Tc,
- the temperature at which heat is received in the generator Tg.
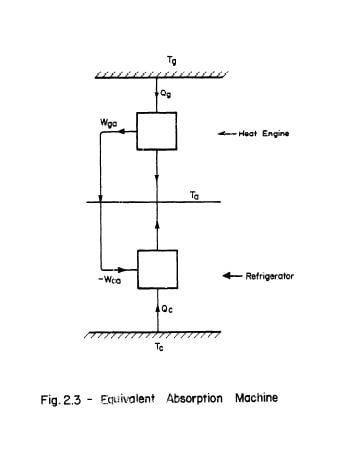
It is possible to imagine an arrangement of reversible machines performing a function equivalent to that of the absorption plant, Fig. 2.3. Firstly, a reversible heat engine receives a quantity of heat Qg, at a temperature Tg and rejects heat at a temperature Ta while producing a quantity of work Wga with an efficiency,
where all temperatures are measured on the thermodynamic temperature scale. Secondly, a reversible refrigerator receives a quantity of heat Qc at Tc and rejects heat at Ta while absorbing a quantity of work Wca. The coefficient of performance of the refrigerator is
If Wga is made equal to - Wca this plant will be equivalent to an absorption refrigerator, The coefficient of performance of the combined plant can be defined as Qc / Qg, which on combining the two previous expressions becomes,
The practical importance of this result is that if a C.O.P. for the cycle under consideration is known Tg may be calculated, since Ta is fixed and Tc is chosen by the designer.
Rigorous Analysis of the Ammonia-Water Cycle
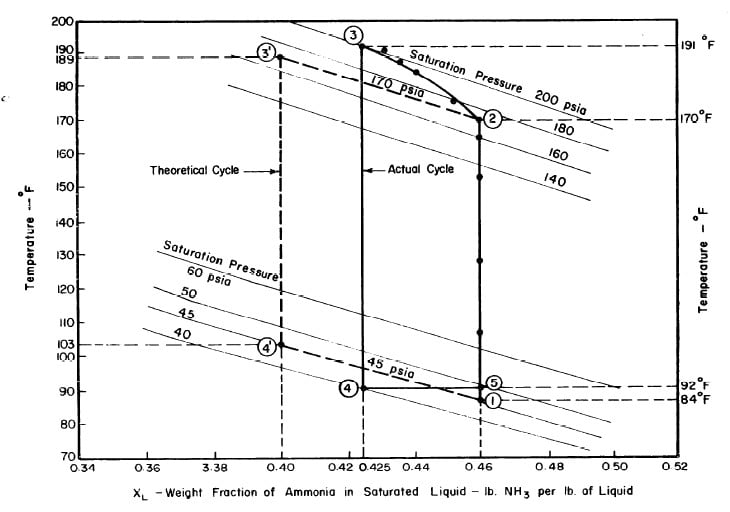
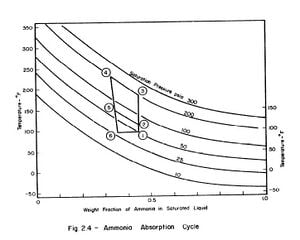
CHINNAPPA (1961), presents a rigorous analysis of the theoretical aqua-ammonia cycle, Two forms of the cycle are suitable for comparison with the actual cycle. These two cycles are shown plotted for the aqua-ammonia system on a pressure-temperature-concentration (p-t-X) chart. The first form of the theoretical cycle may be designated 'constant pressure absorption cycle' and is represented in Fig. 2,4 by 2-3-4-5-2. The second form of the theoretical may be designated 'constant temperature absorption cycle' and is represented in Fig. 2.4 by 1-3-4-6-1.
Even though the constant pressure cycle is the more efficient one it is difficult to realise in practice. Hence, the constant temperature cycle is examined in greater detail.
In the constant temperature absorption cycle regeneration consists of two processes 1-3 and 3-4. In the refrigeration phase during the cooling process 4-6 the solution is cooled, usually by immersion in a water bath, to a temperature t6 which is equal to the initial temperature t1. Effective refrigeration takes place during the process 6-l.
The expression for the amount of refrigeration is
where
- = mean latent heat of the refrigerant during the process 6-1.
- = weight on the refrigerant at point 6.
The heat supplied during the regeneration process 1-3-4 is given by
where
- = weight of the solution, suffix indicating the point of the cycle,
- = specific enthalpy of the solution, suffix indicating the point of the cycle,
- = specific enthalpy of the vapor boiling out of the liquid,
- = differential mass of the vapor boiling out of the liquid.
- Thus the expression for the C.O.P. becomes
Historical Development
According to the Survey of Solar-Powered Refrigeration carried out by SWARTMAN, HA, and NEWTON (1973), the first study undertaken to explore the use of solar energy for refrigeration was probably in 1936 at the University of Florida by Green. The steam to power a steam jet refrigerator was produced by heating water flowing in a pipe placed at the focal line of a cylindro-parabolic reflector.
Oniga reported in 1937 that researchers in Brazil tried to adapt a parabolic reflector to an absorption refrigerator but the system never got beyond the experimental stage.
Kirpichev and Baum of Russia reported the successful operation of an assembly of solar refrigerators producing 250 kilogrammes of ice per day in 1954. The refrigerators were of the conventional vapour – compression type driven by a heat engine operating on the steam produced by a boiler placed at the focus of a large mirror. However, it has been generally conceded that the low efficiency of solar energy in producing power, the very high cost of equipment, and the complexity of this type of system are unfavourable factors in the future development. Since this system was build, there has been little interest shown in this direction of solar refrigeration.
The first major project on an all solar absorption refrigeration system was undertaken by TROMBE and FOEX (1964). Fig. 2.5 shows the general set-up of the system, which has these main features: ammonia-water solution is allowed to flow from a cold reservoir through a pipe placed at the focal line of a cylindro-parabolic reflector. Heated ammonia-water vapourized in the boiler is subsequently condensed in a cooling coil. The evaporator is a coil surrounding the container used as an ice box. The cylindro-parabolic reflector measured 1.5m2. In the prototype trials, the daily production of ice was about 6 kilogrammes or about 4 kilogrammes of ice per square metre of collecting area for four – hour heating.
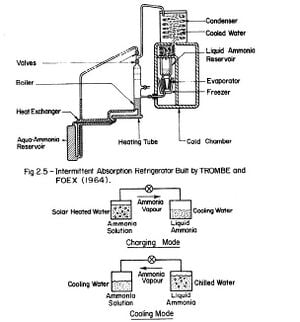
Fig. 2.6 - The Basic Solar-Powered Intermittent Absorption Refrigerator
The design by Trombe and Foex is very promising and should be studied further although modifications may be necessary on the solar collector, boiler, and condenser.
Williams and others at the University of Wisconsin built a small food cooler in 1957 intended for use in underdeveloped rural areas. The apparatus consisted of two vessels linked together by a pipe as shown in Fig. 2.6. The energy was provided by a parabolic mirror of moulded 1.27 mm polystyrene with an aluminized mylar polyester film and stiffened at the rim by metal tubing. Ammonia-water and R-21-glycol ether were used as working solutions. This study showed that refrigeration can be achieved by the use of intermittent absorption refrigeration cycles. Although performance is limited by the characteristics of the intermittent cycle, the simplicity of the system accounts for the low temperature obtained in the evaporator. Finally, the study showed that ammonia-water has a superior performance over R-21-glycol ether in an intermittent refrigeration system.
CHINNAPPA (1962) built a simple intermittent refrigerator operated with a flat-plate collector at Columbo, Ceylon as shown in Fig. 2.7. The generator-absorber in this refrigerator was of welded pipe construction and incorporated with a flat-plate collector and a water cooled absorber. The solar collector was a copper sheet measuring 152.4 cm by 106.7 cm, 0.76 mm thick, and painted black. The plate was solded to six 6.35 cm diameter steel pipes and the pipes were welded to headers. There were three glass covers on the collector which were supported by strips of cork board. An ammonia-water solution was used as the working fluid.
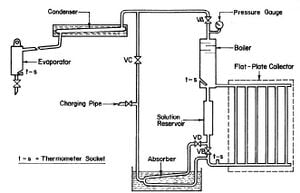
Fig. 2.7 - Schematic of Solar Refrigerator Operated with Flat-Plate Collector by CHINNAPPA (1962)
While it has been generally expected that the flat-plate collector would be more suitable for the lower temperature of generation required in air conditioning, tests in the investigation by CHINNAPPA (1962) indicated that it is possible to use a flat-plate collector incorporated with the generator to produce cooling at a temperature as low as -12°C. It is noted that ice can be produced in this refrigerator at one kg a day per 0.7 m2 of solar collecting surface. Results in this investigation were not spectacular, but they showed that a simple intermittent refrigerator using a low temperature heat collecting device such as the flat-plate collector can achieve cooling.
SWARTMAN and SWAMINATHAN (1971) built a simple, intermittent refrigeration system incorporating the generator-absorber with a 1.4 m2 flat-plate collector at the University of Western Ontario. Fig. 2.8 shows the system schematically. The collector-generator assembly consisted of 1.27 cm steel pipes connecting a 5.1 cm feeder and 15.2 cm header. Thin copper sheet was soldered to the tubes and the whole assembly was enclosed in a wooden box with insulation material at the bottom and a two-layered glass cover on the top. Ammonia water solutions of concentration varying from 58 to 70 percent were tested. Tests were relatively successful; evaporator temperatures were as low as -12°C, but due to poor absorption, the evaporation rate of ammonia in the evaporator was low.
Another study at the University of Western Ontario in 1970 investigated an ammonia-sodium thiocyanate solution in the same system as that described above. Results of the investigation showed that the coefficients of performance for NH3 -NaSCN range from 0.11 to 0.27 compared with 0.05 to 0.14 for NR3-H20 obtained from the earlier study, Nevertheless, the system was
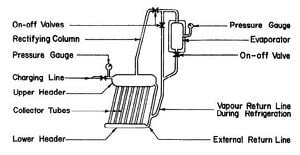
Fig. 2.8 - Intermittent Solar Refrigerator Built at University of Western Ontario
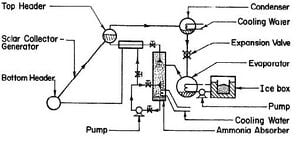
Fig. 2.9 - Solar Ice Maker Built at the University of Florida by FARBER (1970)
still unable to make any considerable amount of ice at the evaporator. It was concluded that NH3-NaSCN has a better performance than that NH3-H20. It also offered lower equipment cost as it did not need a rectifying column due to the low volatility of the NaSCN salt. An optimal concentration of 54% was suggested for intermittent refrigeration.
FARBER (1970) has built the most successful solar refrigeration system to date. It was a compact solar ice maker using a flat-plate collector as the energy source. Fig. 2.9 shows the flow diagram of the system. The solar collector-generator was 1.49 m2, consisting of a 6.35 cm top header. The 2.54 cm pipes were spaced on 10.2 cm centres and soldered to a 20 gauge galvanized iron sheet. This unit was placed in a galvanized sheet metal box with a single glass cover and one inch of Styrofoam insulation behind the absorber-generator element. In addition to the usual components, such as condenser, evaporator, ice box, heat exchanger, there was an ammonia absorbing column of the shell-and-tube type and two pumps to circulate the liquid ammonia and chilled water in the evaporator. It was reported that an average of about 42,200 kJ of solar energy was collected by the collector per day and ice produced was about 18.1 kilogrammes. This gave an overall coefficient of performance of about 0.1 and 12.5 kilogrammes of ice per m2 of collector surface per day.
As far as solar refrigeration is concerned, this has been the most successful system, but it should be noted that the system was not totally solar-powered as there were two pumps operated by electricity. The system would not work in areas where electricity is not available.
III DESIGN OF THE EXPERIMENTAL UNIT
Choice of Configuration
It was stated earlier that a solar refrigerator consists of two components, a solar power unit and a refrigeration unit. The solar power unit is based on either of two basic concepts, i.e., flat-plate collectors or focussing collectors.
Flat-plate collectors are flat blackened surfaces to absorb direct and diffuse solar radiation. Transparent covers and back insulation may be provided to reduce or control heat losses from the plate. On the plate, absorbed solar energy is converted to a desired form of energy, usually heat, and means are provided to remove that energy, usually as heated water or air. Flat-plate collectors are generally suitable for operation in a fixed position.
The basic element of the focussing collector is an optical device, e.g., a parabolic reflector, to focus the beam component of solar radiation on a receiver smaller than the reflector. This collector can produce a higher energy flux. Although the focussing collector gives higher temperatures than the flat-plate collector, it is more difficult to operate. Also, for a small experimental -unit, it seems to be more expensive than the flat-plate collector, Therefore, a flat-plate collector was selected for this particular study.
The refrigeration unit can be either a continuous or an intermittent absorption system. The continuous absorption refrigeration system cannot serve the purpose if the pumps require power. Therefore, in rural areas where electricity is unavailable, the intermittent absorption refrigeration
system is preferred. The intermittent refrigeration cycle has two major operations, regeneration and refrigeration. Regeneration is the process of heating the refrigerant-absorbent fluid to drive off the refrigerant vapour and condense the vapour in a separate container. Refrigeration takes place when the liquid refrigerant vapourizes, producing a cooling effect around the evaporator. The refrigerant is re-absorbed by the absorbent. Since the refrigerator is a purely experimental device it was decided to keep it as simple as possible. The configuration chosen is shown in Fig. 3.1. Simplicity has been achieved by having the condenser function as the evaporator and the generator function as the absorber.
Operation of the System
During the regeneration, valve A is open and valve B is closed, and the strong solution in the generator being heated by the flat-plate collector boils, producing vapour at a high pressure. The weak solution returns from the top header to the bottom header by the insulated return pipes. The vapour in the top header is mainly ammonia because water has a much lower volatility than ammonia. The ammonia vapour passes into the condenser which is immersed in a tank of cold water to keep it cool. The pressure is uniform throughout the system. When heating stops valve A is closed and the vapour pressure in the generator drops. The concentration in the generator is now less than it was before regeneration. Before refrigeration is started the tank of cooling water is removed and valve B is opened. The condenser now functions as the evaporator. Ammonia vapourizes due to the pressure difference between the generator and evaporator. The vapourization of ammonia absorbs heat from
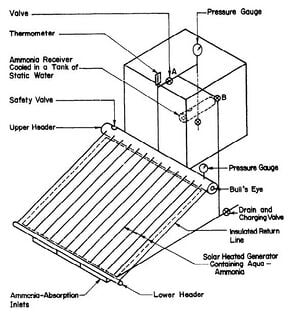
Fig. 3.1 - The First Experimental Unit
the surroundings of the evaporator, thus producing the refrigeration effect. Ammonia vapour from the evaporator passes through the pipe taken to the bottom header of the generator so that the incoming vapour bubbles through the aqua-ammonia solution thus facilitating absorption in it. The glass covers are removed from the collector so that the heat of absorption can be dissipated to the sky from the generator risers. Refrigeration continues until all the liquid ammonia in the evaporator has vapourized. A full cycle of operation has now been completed. To accommodate the intermittent availability of solar energy, the refrigeration is carried out during the day and refrigeration takes place at night after the radiation is no longer available.
Concentration of Aqua-Ammonia
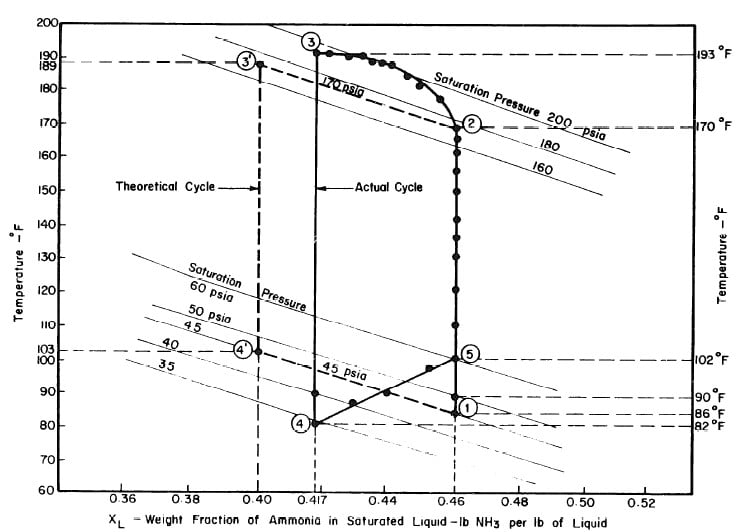
The objective is to produce a temperature of 17°F in the evaporator, The saturation vapour pressure of anhydrous ammonia at this temperature is 45 psia. The temperature of the absorber is the atmospheric temperature which is assumed to be 86°F. Thus in the absorber there is an aqua-ammonia mixture at temperature of 86'F with the pressure of ammonia vapour at 45 psia. Hence, from the p-t-x diagram for aqua-ammonia the concentration is found to be 0.46, thus determining the starting point of refrigeration cycle, shown as point 1 in Fig. 3.2.
Regeneration Phase of the Cycle
The condenser temperature is 86°F. From the p-t-x diagram the saturation pressure of anhydrous ammonia at this temperature is 170 psia. Point 2 of the cycle can be determined, since the pressure and the concentration (which

Fig. 3.2 - Ideal Thermodynamic Cycle
does not change during process l-2) at point 2 are known. Point 3 of the cycle is fixed by the maximum solution temperature attainable with the collector, which is assumed to be 189°F. This determines point 3 and hence the concentration which is 0.40 from the p-t-x diagram.
Refrigeration Phase of the Cycle
Ideally, during the refrigeration phase of the cycle, the solution is first cooled to the absorption pressure of 45 psia, which at a concentration of 0.40 corresponds to an initial absorption temperature of 103°F. This fixes point 4. The cycle is completed by the process 4-l during which ammonia evaporating at 17°F is reabsorbed into the solution.
Collector-Generator Specifications
It was decided to keep the unit as compact as possible. Thus, a four ft. by four frontal area was selected for the collector-generator. Black iron seamless pipes were used throughout to resist corrosion by ammonia-water mixture and the pressure associated with the necessarily high ammonia concentrations. A copper sheet four by four feet and 0.06 inch thick was used for the collecting plate and was painted dull black. The plate was soldered to twelve 1 inch diameter tubes at four-inch intervals. The ends of the 1 inch tubes were welded to headers. To provide for adequate separation of the water from the ammonia vapour out of the collector-generator, a 4-inch pipe was used for the top header, This 56 in, length of pipe gave a liquid surface area of 225 in2 when the header was half full. The liquid level could be observed through bull's eyes at both ends of the header. For the bottom header a pipe 2 inches in diameter and 54 inches long was used. The arrangement of the collector-generator is shown in Fig. 3.3.
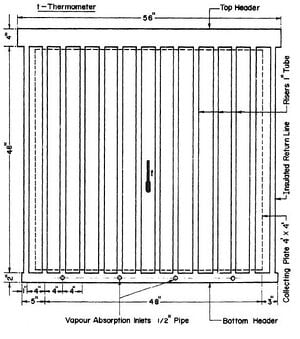
Fig. 3.3 - Solar Collector - Generator
To prevent heat loss at the rear of the collector-generator polystyrene foam four inches thick was used for insulation. The top and bottom headers and the risers at each end of the collector were also thermally insulated with polystyrene foam. There were two glass covers in front of the collecting surface supported by a wooden frame. Ordinary window glass 1/4 in thick was used. The gap between the collecting tubes and the first glass cover was 1/4 in; between the two glass covers the gap was 3/4 in. The glass covers were removable.
The inclination of the plane of the generator was 20 degrees to the horizontal with the unit facing due south.
The Volume of the Generator
The volume of the generator calculated below from the standard pipe dimensions is used to determine the quantity of aqua-ammonia in the system, and to determine the changes in the liquid level in the generator throughout the cycle.
Top header (half full)
- 667 ft x 0.5 x 0.0882 ft3/ft = 0.206 ft3
14 risers 14 x 4 ft x 0.00585 ft3/ft = 0.328 ft3 Bottom header
- 5 ft x 0.0233 ft3/ft = 0.105 ft3
Total volume = 0.639 ft3
Surface area of the liquid in the top header half full
= ID. X length = 4.026 in x 56 in = 225.456 in2 = 1.565 ft2
Specific Volume of aqua-ammonia
At point 1, V1 = 0.0192 ft3/lb
Point 2, V2 = 0.0205ft3/lb Point 3, V3 = 0.0202 ft3/lb Point 4, V4 = 0.01895 ft3/lb
Liquid level in generator Start with 0.639 ft3 of 0.46 aqua-ammonia at 86°F Its weight is 0.639/0.0192 = 33.281 lbs
The volume of 33.281 lbs of 0.46 aqua-ammonia at 170°F is 33.281 x 0.205 = 0.682 ft3
Increase in volume is 0.682 – 0.639 = 0.043 ft3
Rise in liquid level is 0.043/1.565 = 0.027 ft
= 0.331 in
When concentration, X = 0.46
Wt. Of ammonia + wt. of water = 33.281 lbs
Therefore, wt. of ammonia = 15.309 lbs wt. of water = 17.972 lbs
When concentration X = 0.40
wt. of ammonia = 11.981 lbs
wt. of water = 17.972 lbs
Total weight = 29.953 lbs
Therefore, wt. of ammonia distilled = 3.328 lbs.
After the distillation of 3.328 lbs of ammonia we have 29.958 lbs of 0.40 aqua-ammonia at 139°F
Volume = 29.953 x 0.0202 = 0.605 ft3
Decrease in volume below initial volume at point 1 is 0.034 ft3.
Fall in liquid level below centre is 0.034/1.565 = 0.022 ft
0.568 ft3
Decrease in volume below initial volume at point 1 is 0.071 ft3.
Fall in liquid level below centre is 0.071/1.565 = 0.045 ft = 0.544 in.
The Size of the Receiver for Ammonia
Weight of ammonia distilled = 3.328 lbs
This ammonia has volume (at 86°F) = 3.328/37.16 = 0.089 ft3.
Let the ammonia receiver be made of Schedule 40, 4-inch pipe. Required length = 0.089/0.0882 = 1.015 ft = 12.18 in.
Consequently, the ammonia receiver (condenser-evaporator) was made of 4 inch black iron pipe, 16 inches long.
Heat of Generation
Let enthalpy of 29.953 lbs of 0.40 aqua-ammonia at 189°F = H3, enthalpy of 3.328 lbs of ammonia vapour at mean generation temperature (approximately) 178° = HA, enthalpy of 33.281 lbs of 0.46 aqua-ammonia at 86°F = H1.
From fig. 3.2: Hl = 33.281 x (-55) = -1830 Btu.
HA = 3.328 x 627 = 2086 Btu.
H3 = 29.953 x 75 = 2246 Btu.
Therefore, heat of generation = H3 + HA – Hl = 6162 Btu.
Daily global solar radiation on horizontal surface = 400 Cal.cm.-2day -1
= 22,800 Btu on 4 by 4 feet surface per day
Therefore, the solar energy incident on the collector is 3.7 times the heat of generation.
Heat of Condensation
After rectification the ammonia has a temperature of 120°F.
Enthalpy of 3.328 lbs of ammonia vapour at temperature 120°F
= 3.328 x 634 = 2110 Btu.
Enthalpy of 3.328 lbs of ammonia liquid at pressure 170 psia and temperature 86°F = 3.328 x 138.9 = 462 Btu. Total heat of condensation = 2110 - 462 = 1648 Btu.
The condenser was kept at a temperature constant within 1°F by immersing it in 135 gallons (80 x 80 x 80 cm3) of cold water during the generation cycle. The water tank was supported by a wooden stand.
Further Details of the Design
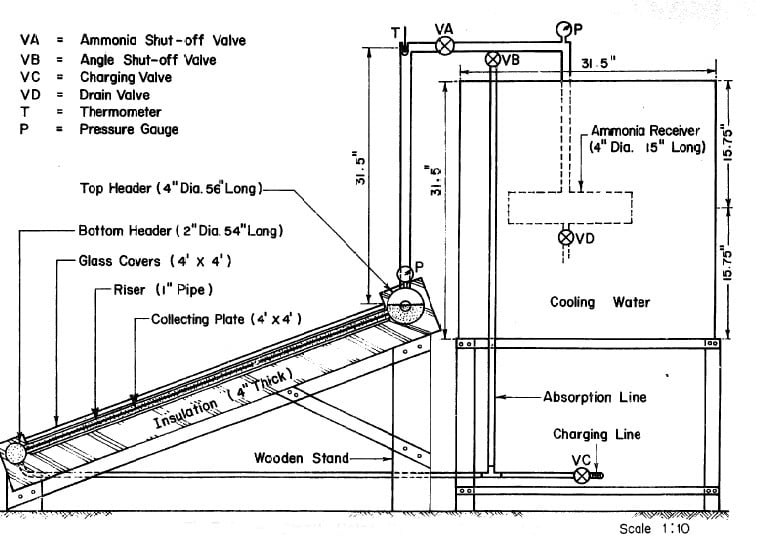
A 1-inch pipe was used to connect the generator to the ammonia reservoir. A 28 in. length of this pipe rising vertically from the top header was used as a rectifier to remove water from the ammonia being distilled. The absorption line was made of ¼ in pipe connected to the bottom header as shown in Fig. 3.4.
Fig. 3.4 - Small Solar Powered Refrigerator
Fig. 3.5 -Solar-Powered Refrigerator
There were two ammonia shut-off valves to control the system. The pressure in the system was indicated by two bourdon-type ammonia gauges; one was attached to the generator and the other was at the top of the tube leading to the ammonia receiver. A thermometer was also used at the top of the rectifier to measure the temperature of the ammonia vapor.
IV EXPERIMENTAL TESTS
Relationship between Plate Temperature and Solution Temperature
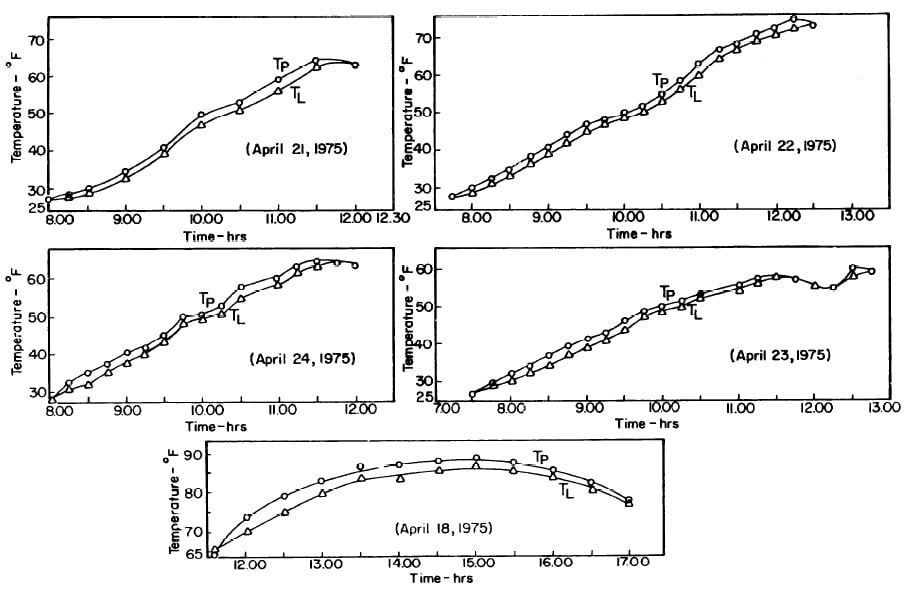
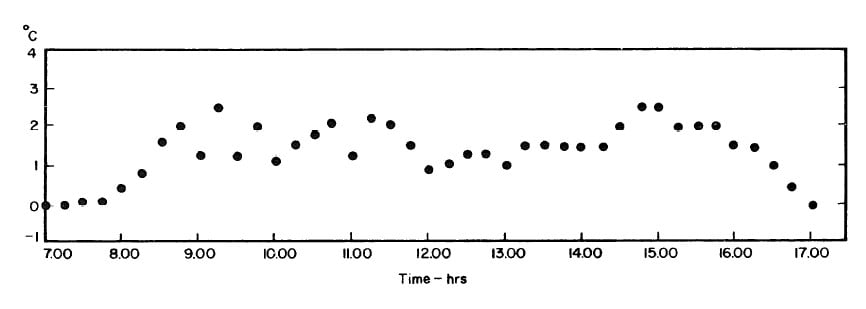
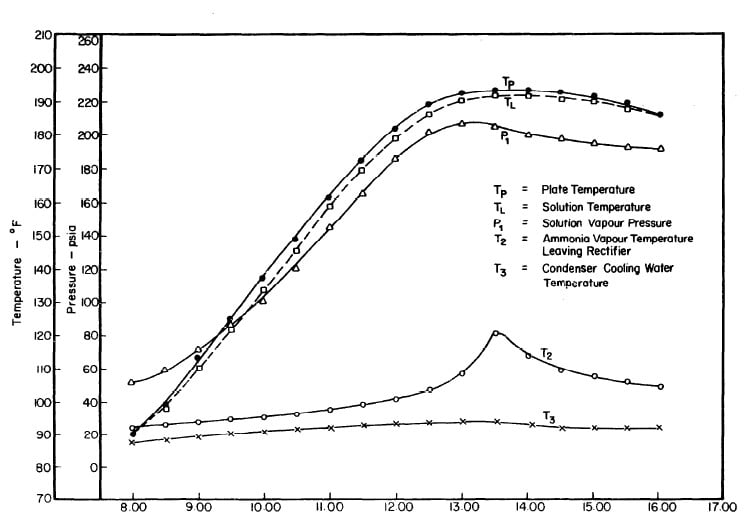
The collector-generator was first charged with water and temperature measurements were made to find the relation between the plate temperature (TT)and solution temperature (TL). Five tests runs were carried out (see Fig.4.1). It was concluded that the solution temperature was lower than the corresponding plate temperature by about 2.4°F. However, it was observed that at the beginning and end of each day both temperatures were the same.The average values of the temperature differences TP-TL are shown in Fig. 4.2.This calibration was necessary because no high pressure thermometer fitting had been attached to the generator for measuring internal temperatures.
Experimental Results
After evacuation, the system was charged with 0,46 aqua-ammonia solution (see Appendix A). The results obtained during four test runs are shown in Figures 4.3 to 4.14. These runs were performed on nearly cloudless days.
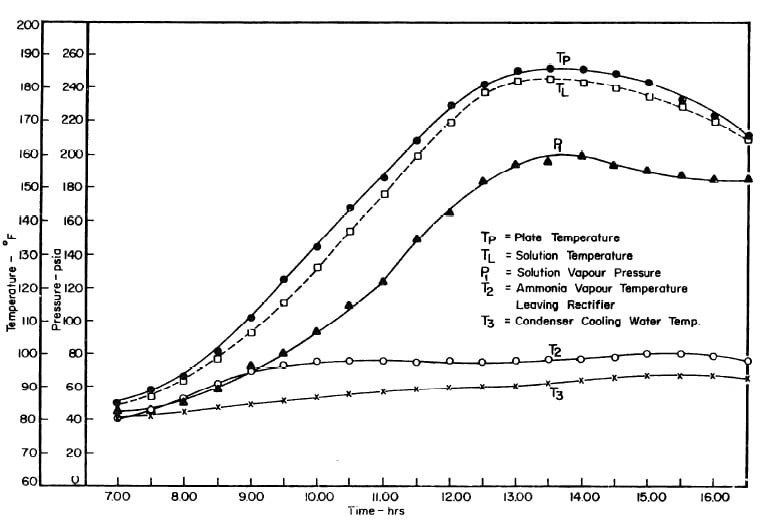
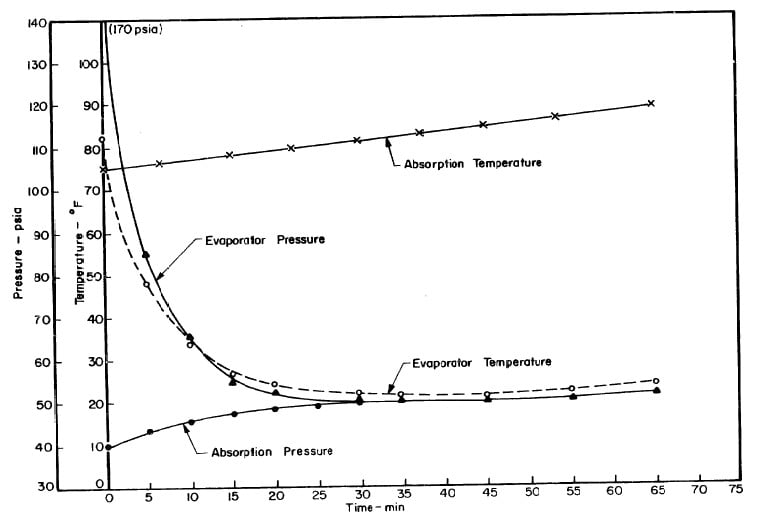
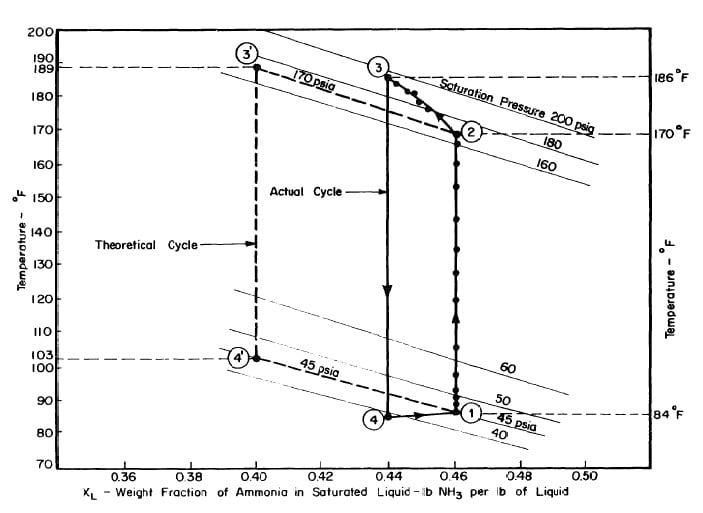
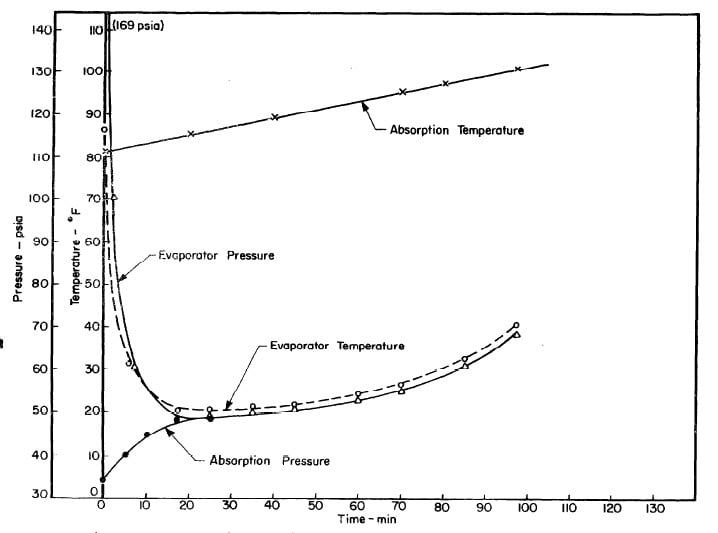
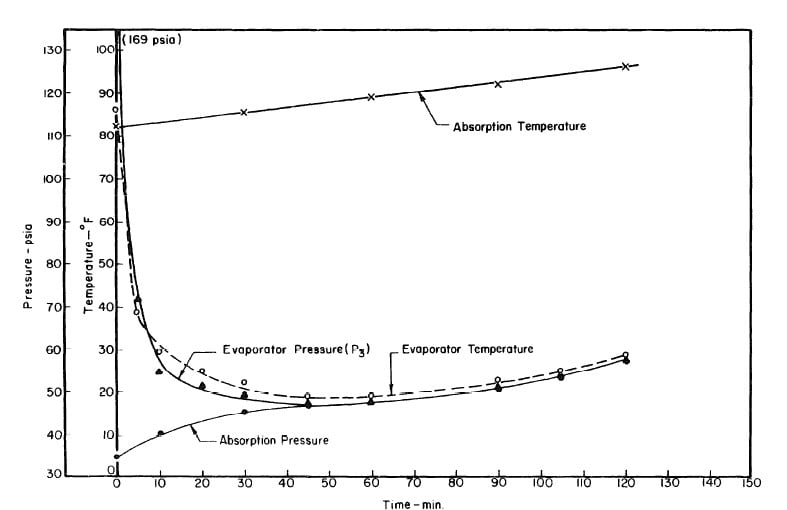
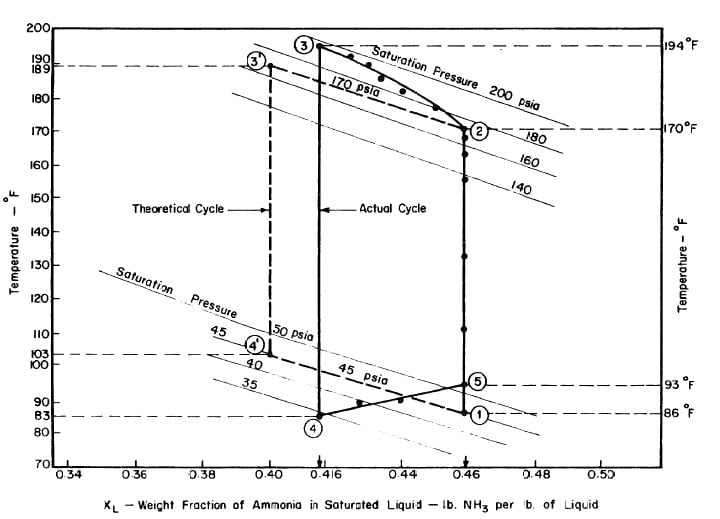
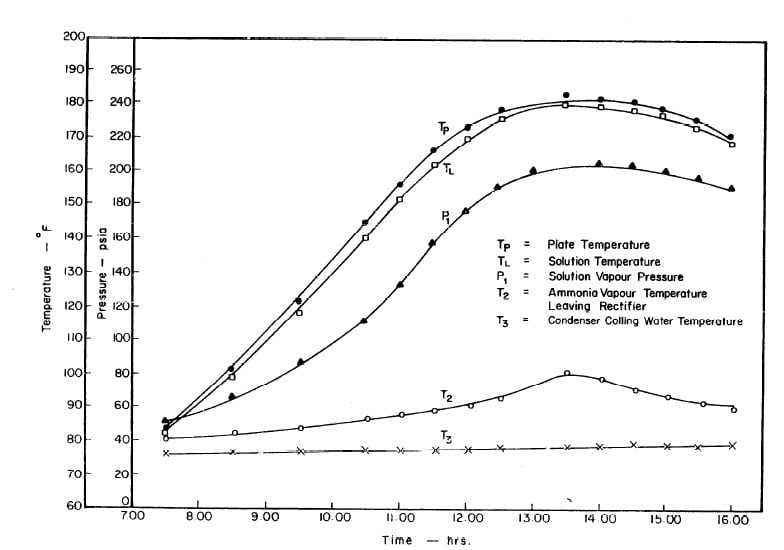
Illustrated in Fig. 4.3 are the plate temperature (Tp), the solution temperature (TL)derived from the calibration shown in Fig. 4.2, the solution vapor pressure (Pl), ammonia-vapor temperature when leaving rectifier (T2),and condenser cooling water temperature (T3) during the generation period. The evaporator pressure, evaporator temperature derived from the pressure, absorption pressure, and absorption temperature for the refrigeration period are shown in Fig. 4,4. The theoretical and actual cycles executed by the solution in the collector-generator are shown as l-2-3'-4' and l-2-3-4-5 respectively in Fig. 4.5.
Fig. 4.1 - Observation on Plate and Solution Temperatures
Fig. 4.2 - Differences between Plate and Solution Temperature (TP-TL): Mean of Five Test Runs.
Fig. 4.3 - Observation during Refrigeration test on May 9, 1975
Fig. 4.4 - Observation during Refrigeration test on May 9, 1975
Fig. 4.5 - Actual and Theoretical Solution Cycles for Test on May 9, 1975
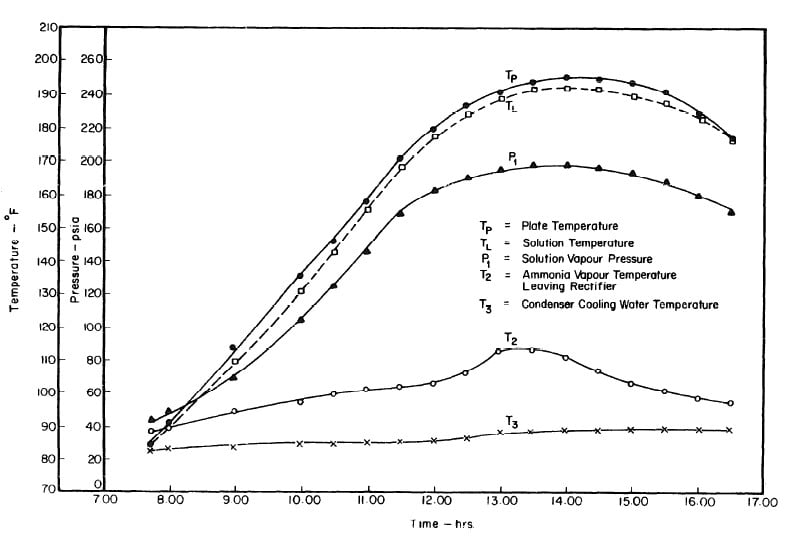
Fig. 4.6 - Observations during Regeneration Test on May 10, 1975
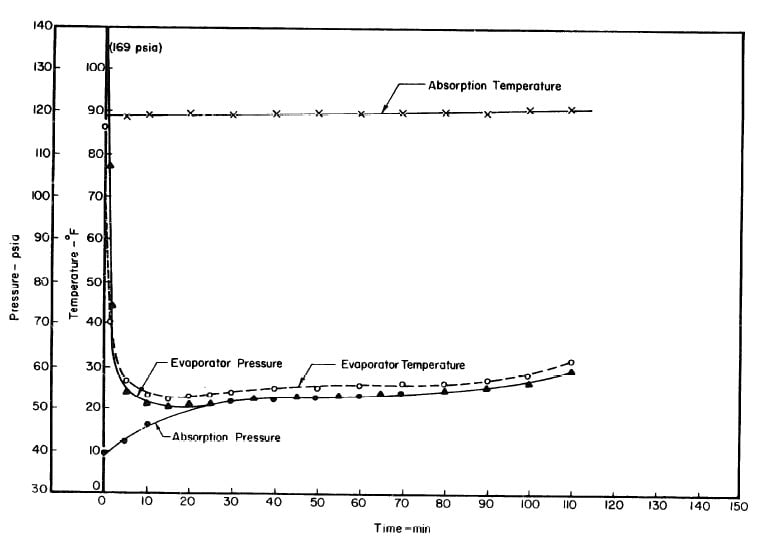
Fig. 4.7 - Observations during Refrigeration Test on May 10, 1975
Fig. 4.8 - Actual and Theoretical Solution Cycles for Test on May 10, 1975
Fig. 4.9 - Observations during Regeneration Test on May 14, 1975
Fig. 4.10 - Observations during Refrigeration Test on May 14, 1975
Fig. 4.11 - Actual and Theoretical Solution Cycles for Test on May 14, 1975
Fig. 4.12 - Observations during Regeneration Test on May 17, 1975
Fig. 4.13 - Observations during Refrigeration Test on May 17, 1975
Fig. 4.14 - Actual and Theoretical Solution Cycles for Test on May 17, 1975
The analysis of the test on may 14th 1974 9Figure 4.9,4.10, and 4.11) is given as an example below.
Amount of Ammonia Distilled
Initially we have:
- Concentration of solution = 0.46
- Total weight of solution = 33.281 lbs
- Weight of ammonia = 15.309 lbs
- Weight of water = 17.972 lbs
After regeneration the final concentration of the solution in the collector-generator is 0.416, as shown in Fig. 4.11.
Since
- The weight of water = 17.972 lbs,
- Weight of ammonia in solution = 12.800 lbs.
Therefore
- Amount of ammonia distilled = 2,509 lbs.
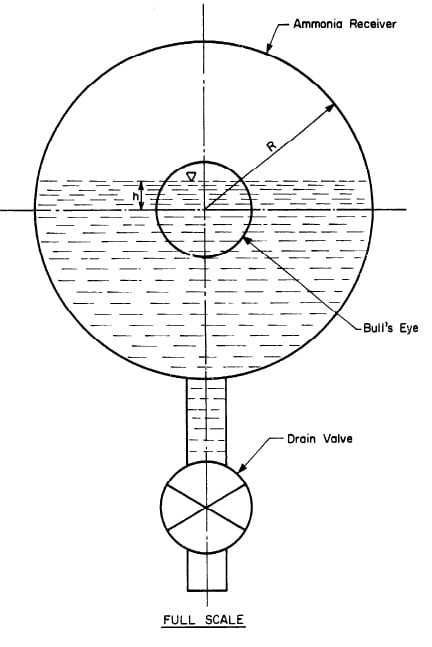
The amount of ammonia distilled was also determined by observing the liquid level in the receiver. Fig. 4,15 shows the geometry of the cross section, of the receiver.
Let
- A be the cross section area of the liquid,
- R be the radius of the receiver cross section,
- h be the height of the liquid level above the center of the receiver,
- 1 be the length of the receiver;
Also, let v be the volume of the drain pipe below the receiver. Then the volume of the liquid is equal to Al + v.
Fig. 4.15 - Cross-Section Ammonia Receiver.
Where
We have R = 2.013 inches, 1 = 1.25 ft, and v = 0.00105 tuft; and after this run h was observed to be 0.3 inches. This gives
- Volume of liquid ammonia distilled = 0.0666 tuft
This volume of liquid ammonia was observed at 7.00 am in the morning after the regeneration; the vapor pressure of the ammonia was 169 psi.
We now have from the ammonia tables:
- Ammonia temperature = 86°F
- Density of liquid ammonia = 37.16 lb ft3
Therefore, weight of liquid ammonia distilled = 2.48 Ibs.
This is in good agreement with the quantity 2.509 lbs previously calculated from the change in the ammonia-water solution. If 2.48 lb of ammonia is distilled, the final concentration in the generator is 0.4165, this confirms the actual thermodynamic cycle as shown in Fig. 4.11.
Cooling Ratio
The cooling ratio of the cycle measures the performance of the system and is defined as
Where
- Qc = cooling available during refrigeration period, and
- Qg = heat absorbed by collector generator during regeneration.
The cooling available during refrigeration period can be calculated as colows.
- 2.509 lbs of liquid ammonia at 86oF (169.2 psia) has enthalpy
- = 2.509 x 138.9 Btu
- = 3.48.9 Btu
- 2.509 lbs of ammonia vapour at 19oF has enthalpy
- = 2.509 x 617.5 Btu
- = 1549.30 Btu
Therefore, cooling obtainable
- = 1540.30 – 348.50
- = 1200.8 Btu
Heat Absorbed by Solution During Regeneration
Let enthalpy of 30.772 lbs of 0.416 aqua-ammonia
- at 193oF = H3,
enthalpy of 2.509 lbs of ammonia vapour at mean generation temperature
- at 180oF = HA,
and enthalpy of 33.281 lbs of 0.46 aqua-ammonia
- at 86oF = H1
From Fig. 4.11,
- H1 = 33.281 x (-55) = -1830 Btu
- HA = 2.509 x (625) = 1568 Btu
- H3 = 30.772 x (79) = 2431 Btu
Total heat absorbed by solution
- = H1 +HA + H3
- = 5829 Btu
Therefore, cooling ratio
- = 1200.8/ 5829
- = 0.209
Solar Coefficient of Performance
The solar C.O.P. is defined as the ration of the cooling obtainable to the amount of solar energy absorbed by the collector plate. The amount of solar energy absorbed by the collected plate can be calculated as shown in detail in Appendix B. For the run on May 14th the amount of solar energy absorbed by the plate
- = 13,237 Btu.
Therefore, solar C.O.P.
- = 1200.8/ 13,237
- = 0.0907
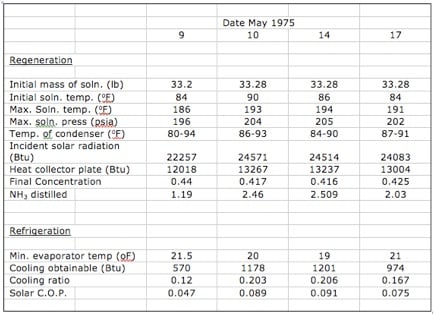
The results of all four experimental runs are summarized in Table 4.1.
Table 4.1 Summary of Experimental results
Fig. 4.16 – Refrigeration Process – Lower Photograph shows frost on the evaporator.
Discussion
Although the system has worked, the cooling ration and the solar C.O.P. are still low as in the previous studies of CHINNAPPA (1962) and SWARTMAN and SWAMINATHAN (1971). It is difficult to control heat losses in the system. However, while Swartman found the absorption process to be slow, there were no such difficulties in the refrigeration process in this system. The absorption process was completed within two hours and the formation of ice on the outer surface of the evaporator took half an hour (Fig. 4.16).
V CONCLUSIONS AND PLANS FOR CONTINUING RESEARCH
Conclusions
The capability of AIT in the design, construction, and operation of solar powered refrigeration has been demonstrated. Furthermore, the operating conditions were found to be almost exactly in accordance with the design specifications. The theory of the system is therefore well understoof. The new feature by whuch ammonia vapour from the evaporator is taken to the bottom header of the generator so that the heat of absorption during the refrigeration process is dissipated from the flat plate has been shown to remove the difficulty encountered by previous workers of obtaining sufficiently rapid absorption for satisfactory operation
Economic Considerations
The cost of making this experimental unit was 15,500 bahts. If annual depreciation and maintenance is 10 percent of the cost, then the cost per day is 4 bahts, the cooling effect obtained on a good day is sufficient to make 2 kilogrammes of ice, and studies of the solar radiation climate show that over one year the average yield would be about 1 kilogramme of ice per day. Therefore, 1 kilogramme of ice would cost 4 bahts. This is eleven times the wholesale price of ice in Bangkok. (0.375 bahts per kilogramme). However, the objective in making this experimental unit was merely to demonstrate the refrigeration effect produced from solar energy, and to gain practical experience; no attempt was made to optimize the performance of the system or to minimize the cost. It seems, therefore that an economically viable solar ice maker is within striking distance.
Modifications
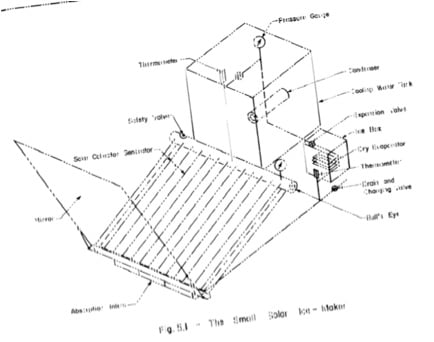
Work is at present under way to test two new features on the refrigerator. The first is an expansion valve with a dry evaporator coil connected between the ammonia receiver and the absorption inlets. The evaporator coils will be used to cool a box for making ice. The second feature is a flat mirror used to enhance the solar heating of the generator. Various positions of the mirror attachment will be tested. These two new features are shown in Fig. 5.1.
The Development of a Village Ice-Maker
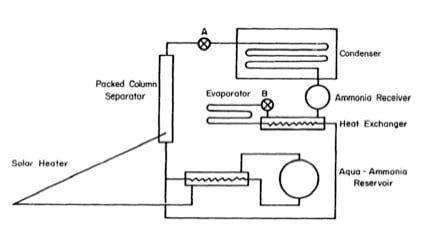
A solar ice maker may be designed for domestic or village use. The larger village sized units would be more efficient and hence relatively cheaper. The main objective will therefore be to design, construct, and test a solar ice maker that produces 100 kilogrammes of ice per day without using oil or electricity. It must be rugged and easy to operate. A unit producing 100 kilogrammes of ice per day requires a solar collecting surface of about 20 square metres. The efficiency of the system will be improved in several ways. High generation temperatures will be avoided by keeping the ammonia concentration in the generator constant with the help of a reservoir containing excess solution. The thermal capacity of the solar heater will be reduced by using a packed column separator instead of a larger diameter header. Heat exchangers will be used to save heat during regeneration and to save cold during refrigeration. The system is shown in Fig. 5.2.
Fig. 5.1- The Small Solar Ice-Maker
Fig. 5.2 – Proposed New Solar Powered Ice Making System
In the daytime, during regeneration, valve A is open, and B is closed. Strong solution from the top of the reservoir passes through the heat exchanger to the bottom of the heater, and weak solution returns to the bottom of the reservoir. Ammonia vapour from the separator is condensed in a coil immersed in cold static water, and the ammonia liquid is collected in the receiver. At night, during refrigeration, valve A is closed, and B is open. Ammonia from the receiver passes through the heat exchanger, the expansion valve B, and evaporator. The vapour is then absorbed in weak aqua-ammonia from the bottom reservoir. The heat of solution is dissipated by the solar heater, and the strong solution returns to the top of the reservoir. Also, the static water surrounding the condenser is cooled by exposure to the night sky. In this system the workability of all the individual units has been reported in the literature, but they have never been combined in this way before. However, the system is unlikely to present any serious technical problems.
Alternatives
It has been recently reported by GUPTA (1976) that R.L. Datta and his group are developing an ammonia-sodium thiocyanate system capable of producing 75 kg of ice per day. The design coefficient of performance is three times the previous obtained value and the problems of rectification inherent in ammonia-water systems will be avoided. A cylindrical parabolic collector 25m2 will be used for heating the generator.
DATTA (1976) himself has remarked that
"It is much easier to cool to 50oF or 60oF and provide a decreased humidity than it is to refrigerate food to ice temperature……… Intensive research should be directed toward inexpensive indigenous equipment to provide such cooling in cellars which might be partly underground. Stationary flat plate collectors with selective surfaces could be used rather than movable focusing collectors, and operation by manpower of the developing countries in their rural areas will be cheaper than automatic machines which require larger capital investment. There is a challenging problem of operating such solar coolers in remote areas where electricity is not available."
The remarks made above show that the development of solar powered refrigeration technology for the use in developing areas is an active field, and that there are various lines of research to be followed. AIT will keep in touch with these developments and is capable of making significant contributions to the field.
References
ANON., (1963), A case for Solar Ice Maker, Solar Energy 7 p.1
BA HLI, F., et al (1970). Possibilities for Solar Ice Makers. International Solar Energy Society Conference, Melbourne, Australia.
CHINNAPPA, J.C.V., (1961), Experimental Study of the Intermittent Vapour Absorption Refigeration Cycle Employing the Refrigerent-Absorbent Systems of Ammonia-Water, and Ammonia-Lithium Nitrate, Solar Energy 5 pp. 1-18.
CHINNAPPA, J.C.V., (1962), Performance of an Intermittent Refigerator Operated by a Flat-Plate Collector, Solar Energy Vol 6 pp. 143-150.
DATTA, C.L., (1976), Solar energy –Its Relevance to Developing Countries. Working Paper, Expert Working Group on the Use of Wind and Solar Energy, March 1976, Natural Resources Division, ESCAP, Bangkok.
FARBER, E.A., (1970), Design and Performance of a Compact Solar Refrigeration System, Paper No. 6/58, 1970, International Solar Energy Society Conference, Melbourne, Australia.
GUPTA, C.L., (1976), Solar Energy in India, Working Paper, Expert Working Group on the Use of Wind and Solar Energy, March 1976, Natural Resources Division, ESCAP, Bangkok.
MERRIAM, M.F., (1972), Decentralized Power Sources for Developing Countries, Technology and Development Institute East-West Centre, Working Paper Serious NO. 19
NATIONAL ACADEMY OF SCIENCES (1972), Solar Energy in Developing Countries: Perspectives and Prospects. National Academy of Sciences, Washington, D.C.
SWARTMAN, R.K., HA, V.H., NEWTON, A.J., (1973) Survey of Solar-Powered Refrigeration, the American Society of Mechanical Engineers, August 1973, 73-WA/Sol-6
SWARTMAN, R.K., and SWAMINATHAN, C., (1971), Solar Powered Refrigerator, Mechanical Engineering, June 1971, Vol. 6, pp. 22-24.
Trombe, F., and FOEX, M., (1964), Economic Balance Sheet of Ice Manufacture with and Absorption Machine Utilizing the Sun as the Heat Source, New Courses of Energy, Vol. 4, U.N. Publication Sales No. 63.I.38, pp. 56-59
Appendix A
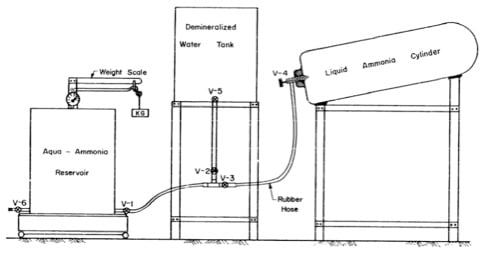
Charging
For this research, a solution of ammonia in water was required. It is sometimes more economical to buy anhydrous ammonia and unload directly into water under controlled conditions to obtain a solution of the desired strength. Since the solar-powered refrigerator was a closed-system, it did not require periodic recharging. Recharging was required if the unit developed a leak or if the ammonia was bled off for any reason.
Equipment
- 1. Liquid ammonia cylinder
- 2. Demineralized water tank
- 3. Aqua-ammonia reservoir
- 4. Pressure gauge
- 5. Rubber hose
- 6. Potassium Dichromate
- 7. Weighing scale, 0-200 lbs
- 8. Valves
- 9. Vacuum pump.
Procedure
Set up the equipment as shown in Figs. A1, and A2. It is necessary to remove air from the system. the presence of air impairs the performance of the system. Therefore, a vacuum pump is required. It is advisable to add potassium dichromate (1 ounce per 60 pounds) to the demineralized water to minimize internal corrosion of the solar-refrigeration system.
The following steps are taken in the charging procedure:
- a) Open V-6, V-1, V-2, V-3 to evacuate air from the system by a vacuum pump, then close all valves.
- b) Weigh the empty aqua-ammonia reservoir.
- c) Open V-1, V-2 and V-5 to let 18.9 pounds of water into the aqua-ammonia reservoir; close V-5 and V-2.
- d) Open V-3 and V-4 gently to let 16.1 pounds of liquid ammonia slowly into the reservoir, the close V-3 and V-4.
- e) Allow the reservoir to cool for about six or eight hours after which the pressure will fall to a low level.
- f) Evacuate the solar powered refrigeration unit by a vacuum pump.
- g) Remove the aqua-ammonia reservoir from the mixing equipment and fit it to the refrigeration unit as shown in Fig. A3.
- h) To get charging flow from the aqua-ammonia reservoir to the generator the vapour pressure in the reservoir must be higher than the vapour pressure in the generator. Therefore the generator is cooled by watering and the reservoir is heated by the sun.
- i) The charging process is stopped when the upper header of the generator is half full.
Fig A1 –Mixing Equipment
Fig. A2 – Mixing Equipment
Fig. A-3 Charging Equipment
Appendix B
Estimation of Incident Solar Radiation
It is assumed that the diffuse radiation in May in Bangkok is 200 calcm-2day-1. We shall also assume that the diffuse radiation on an inclined plane is a linear function of the angle of inclination and is one half of the maximum amount when the plane is vertical. The angle of inclination = 20 degrees to the horizontal. Therefore, the diffuse radiation on the inclined collector (D') is estimated to be 200 – 20/90x100 = 178 calcm-2day-1. Daily total global radiation at the Asian Institute of Technology were recorded using a Bimetallic Actinograph installed on the roof of the north engineering building of AIT. The results are shown in Fig. B1. During the period of the tests the sun passed very close to the zenith at midday. It was therefore assumed as an approximation that, since the collector (which paced south) was inclined at an angle of 200 to the horizontal, the vertical component of direct solar radiation should be multiplied by a factor cos 20o in order to estimate the component of direct radiation normally incident on the collector. Since global radiation (Q)
- = direct radiation + diffuse radiation.
Total radiation on the inclined collector (Q')
- = D' + (Q-200)cos20o
- = 178 + (Q-200)x0.94 calcm-2day-1,
where Q is the daily global radiation.
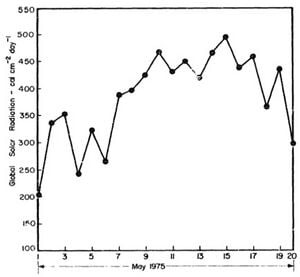
Fig. B1 – Daily Global Solar Radiation at the Asian Institute of Technology
for the test run on May 14, 1975
- Q = 467 calcm-2day-1.
Therefore,
- Q = 429 calcm-2day-1
- = 24,514 Btu.day-1 on 4'x4' collecting area
- = 16.342 Btu per 8 hours
Assume the transmission by the glass cover = 90 percent, therefore, about 90 percent of the original incident solar energy will be transmitted by the first glass cover and another 90 percent by the second glass cover.
Therefore, the heat that proceeds to the collector plate is about 13,237 BTU.
It should be emphasized that the estimates obtained by this method are very rough.
Further Readings
solar refrigeration for vaccines
| Authors | R.H.B. Exell, Sommai Kornsakoo, D.G.D.C Wijeratna, Jianlang Mai, Scott Gennings, Graham Coote, Ana Herrera, Howard Swartz |
|---|---|
| License | CC-BY-SA-3.0 |
| Organizations | Queen's University |
| Cite as | R.H.B. Exell, Sommai Kornsakoo, D.G.D.C Wijeratna, Jianlang Mai, Scott Gennings, Graham Coote, Ana Herrera, Howard Swartz (2010–2025). "The Design and Development of a Solar Powered Refrigerator". Appropedia. Retrieved May 31, 2026. |