Fertilizers

Fertilisers are substances that can be supplied to the soil so as to improve the soil quality and promote the growth of any plants grown in this soil. Fertilisers come in several types and correct application differs depending on this type. Differences on application may include: method of introducing the fertiliser into the soil, the time of the year when the fertiliser is administered, etc...
There's actually little doubt that fertilizers harm ecosystems. But is this inevitable, and what are the alternatives? Limited use and precise application reduce the effect of eutrophication on waterways. More recent discoveries, e.g. the role of soil fungi, the impact of compost teas, and terra preta, show that there may be much greener ways to create abundance in food production.[verification needed] However, this knowledge is still in its early years - the knowledge is still being developed, and the valuable knowledge that already exists has not yet spread widely.
Chemistry
[edit | edit source]A plant absorbs CO2, H20, minerals (main elements: N, S, K, Ca, P, Mg,trace elements: Fe, Zn, Cu, Mn, Mo, B, other:Cl, Na, Si, Al)
| Element | Forms of absorbing |
| N | No3- (nitrate), Na4+ (ammonium) |
| S | So42- (sulfate) |
| K | K+ |
| Ca | Ca2+ |
| Mg | Mg2+ |
| P | H2PO3- |
fosfates, sulfates, nitrates are thus the forms in which the elements are absorbed Fosfates and gelates are also used as agricultural fertiliser !
Especially N, P, K, Mg are added as fertiliser. The trace elements are usually present in sufficient quantities, but the soil is usually badly equipped to let the plant absorb the elements
No3-, Na4+, So42-, K+, Ca2+, Mg2+, H2PO3-
- in soil water (solution)
- clay-humus complex -->holds positively-charged elements, rain water can not flush these to greater depths
- mineral store
- organic store
Nitrogen (N)
- absorbed from nitrate No3- (immediate absorption), and ammonia Na4+ (conversion takes 3-4 weeks to 2-3 months),
- absorbed from artifical fertiliser as lime nitrate (Ca(NO3)2), ammonium nitrate (NH4NO3, ammonium sulfate ((NH4)2, SO4)
organic material causes more nitrogen problems
Potassium (K): Has a relation to nitrogen; even ratio N-K needs to be respected !
Too much potassium gives problems with magnesium
Potassium is stick to clay-humus complex
(Too much/little ?) Calcium: usually provides a problem with the watertransport inside the plant
| Nitrogen | Products | Nutritional substances | Soil improvement |
| N-poor | Peat | none | improved |
| N-poor | green fertiliser | little | improved water capacity |
| N-poor | compost | little-medium | added humus |
| cow manure | N,P,K | soil structure improved | |
| N-rich | chicken manure | N | little or none |
| N-rich | dried types of manure | N, P, K | very little |
General working with fertilisers
[edit | edit source]Fertilisers have "formula's on them that explain what's in it. This is important for fertilising the soil precisely (so that certain elements are not given too much and/or others too little). Bother overfertilising the soil regarding specific elements as well as underfertilising the soil regarding specific elements can hinder the growth of crops.
Always ask a soil analysis before commencing to fertilise ASEF --> can be contacted for soil-analysis sets (belgium) personal mobile soil analysis tests --> are only capable of determining PH,...
A formula looks like: N-P-K-Mg (12-10-18-5(2)) in percentage
0,6kg N/Are: 100kg ->12 kg (12%)
0,6kg X 100/12= 5 kg
Organic fertilisers: never exceed by more than 10% per element (expensive fertiliser if bought)
acid binding value (ABV)
CaO -> Co(OH)2 (calcium oxide)
1 ABV= 1kg of CaO/100kg lime
Maximum= 5 ABV/Are/year
Burying of green manure: maximum 20 cm deep (with a 2cm thick layer). A similar depth is probably also useful for other fertilisers. In order to dig in the fertiliser at this depth, tillage may be needed.
Types of fertilisers
[edit | edit source]Natural fertilisers
[edit | edit source]- Organic matter/Green manure (unprocessed matter/unprocessed plant matter; whole plants)
- Compost/Humus (plant matter/feces in progress of processing/processed plant matter/feces)
- Compost tea (made with green manure)
- Animal manure/guano (unprocessed, is processed to humus in the soil)
- Night soil (unprocessed human manure, can cause illness so is banned in most countries)
- Bokashi liquid fertiliser (made from any kitchen scraps)
- Fermented animal/human urine
- Fermented manure/water mix
- Fermented plant/water mix
- Biochar
- Leaf Mold
- Limestone
- Seaweed
Chemical fertilisers
[edit | edit source]Overview of the different types
[edit | edit source]Organic fertilizers
[edit | edit source]Organic fertilizers are important as they:
- Improve the structure of the soil. On heavier soils de the adhesion of the particles is reduced so that the manipulatability is improved. On light soils, the adhesion between the particles is increased so that a stable crumb structure is obtained.
- Increase the water-holding capacity of the soil
- Promote soil life: soil organisms use organic materials as food
- Contain plantfeeding elements: the mineralization occurs slowly and is spread over several years (up to 3 years after administration), thus only a part is directly absorbable by the plant. The organic nitrogen compounds are first converted to ammonia and then further converted by microorganisms to nitrate, which is absorbable by the plant.
Soil improvers
[edit | edit source]As soil improvers, the following are usable:
- Compost
- Stable manure
- Green manure
These soil improvers also contain plant food, but in many cases it will be necessary to also use concentrated fertilizers. In the private garden, we can best work with concentrated, organic, slow-acting fertilizer like blood meal, bone meal, horn meal instead of fast-acting fertilizers.
A. Compost
[edit | edit source]Good compost is a living whole of a variety of bacteria and other organisms that constantly convert plant waste into humus. Compost is ready for sale at a garden center, but it is also well possible to make it yourself.
B. Stable manure
[edit | edit source]Compost is a better soil improver than stable manure, especially on more acid soils. Fresh manure from the stables will contain many nutrients (especially N) and too fresh organic material. Therefore, the manure can best be left to settle. We make good old manure by mixing cow manure (main component) with an other type of manure, compost and organic material. We place this in a pile and cover it with a finger-thick layer of soil. After 3 months, the manure is ready for use. Old manure contains, similar to compost, a rich whole of soil organisms and brings these, besides nutrients, into the soil.
C. Green manure
[edit | edit source]To protect the soil organisms in the winter, the soil is best kept covered. For this purpose we sow green manure such as winter rye or ryegrass in late summer that we then dig under in the spring. It also acts as a catch crop for the nutritional elements present in the soil. The green manure takes up the nutritional elements from the soil and it will release these in the spring, after mineralization (especially N). We can also sow leguminous plants (such as clover, vetches,...) in early spring and dig these under later-on in early spring. The root nodules gather N-gas from the air and lock it in the soil. This is especially interesting for leafy vegetables.
Concentrated organic fertilizers
[edit | edit source]The shortage in certain nutrients can often not be remedied with the use of compost and old stable manure. We will then need to resort to fertilizers that contain specific nutrients in higher concentrations.
| Name | Nutrients | Remarks |
| Bone meal | 5% nitrogen, 15% phosphorus and lime | Quantities vary |
| Byobact | trace elements | |
| Cacao waste | 8-10% acid binding constituents | Suitable for calcium-poor soils |
| Dendrovorm | 7% nitrogen, 5% phosphorus and 5% potassium | |
| Dolomitic lime | 20% carbonic magnesium and 30% carbonic calcium | |
| Guano | 14% nitrogen | |
| Hoof meal | 13-14% nitrogen | Works slower than blood meal |
| Blast furnace silica lime | 50% acid binding constituents | Suitable for acidic soil |
| Maerl | 45% acid binding constituents, 6% magnesium and trace elements | |
| Magnesite | a lot of magnesium | Quantity varies |
| Natural phosphate | 13% phosphorus | |
| Serpentine | rich in magnesium, silica and trace elements | Composition varies |
| Thomas slag meal | 3% magnesium, 17% phosphorus and calcium | |
| Feather meal | 12 to 14% nitrogen |
Manure, compost and wastes
[edit | edit source]| Per 100 kg | nitrogen (in kg) | potassium (in kg) | phosphoric acid (in kg) | calcium (in kg) | organic matter (in kg) |
| potato foliage | 0,6 | 0,8 | 0,18 | 1,0 | 20 |
| brown coal ashes | |||||
| 1,0 | 0,6–1,5 | 16,0 | |||
| core wood ashes | |||||
| 10,0 | 3,4 | 30,0 | |||
| conifer tree wood ashes | |||||
| 6,0 | 2,5 | 35,0 | |||
| coal ashes | |||||
| 0,1–0,5 | 0,8 | 3,5–8,5 | |||
| peat ashes | |||||
| 1,0 | 1,2 | 15–30 | |||
| basalt meal | |||||
| 1,51 | 0,87 | 12,63 | |||
| bone meal | 4,0 | 0,2 | 21,0 | 31,0 | |
| cesspit sludge | 0,36 | 0,15 | 0,16 | 0,11 | 15–10 |
| cesspit sludge (with peat) | 1,0 | 0,25 | 1,1 | 0,06 | 20-30 |
| leaves | 1,0 | 0,29 | 0,22 | 1,41 | 85 |
| blood meal | 15,0 | 0,7 | 1,3 | 0,8 | 60 |
| pine and spruce needles | 0,9 | 0,13 | 0,2 | 1,6 | 80 |
| pigeon manure | 1,76 | 1,0 | 1,78 | 1,6 | 30 |
| duck manure | 1,05 | 0,63 | 1,4 | 1,7 | 26 |
| egg shells | traces | ||||
| 40 | |||||
| goose manure | 0,55 | 1,0 | 0,55 | 0,85 | 25 |
| (Stinging nettle ?) extract | 0,25 | 0,5 | 0,01 | 0,03 | 5 – 8 |
| plaster | |||||
| 35,0 | |||||
| green manure | yield luguminous crops: 150–250 kg/ha | ||||
| vegetable waste | 0,4 | 0,5 | 0,2 | 0,5 | 30 |
| soil compost | 0,02 | 0,15 | 0,15 | 0,6 | 8 |
| Fabaceae | 0,7 | 0,5 | 0,1 | 0,3 | 20 |
| wool waste | 3–9 | 0,1 | 0,5 | 0,5 | 85 |
| soapy water | |||||
| 0,6 | |||||
| sea sludge | 0,35 | 0,7–0,92 | 0,2–3,5 | 6–8,12 | 40 |
| sea weed | 0,19 | 0,29 | 0,04 | 0,54 | 5 |
| hair | 3-9 | ||||
| present | |||||
| hoof chips | 11,0 | ||||
| 6,0 | 6,6 | 80 | |||
| horn meal | 10,2 | ||||
| 5,5 | 6,6 | 85 | |||
| horn chips | 17,0 | ||||
| 8,0 | 6,6 | 85 | |||
| wood chips | 0,1 | 0,003 | 0,001 | ||
| 80 | |||||
| chicken manure | 1,63 | 0,85 | 1,54 | 2,4 | 26 |
| cow manure | |||||
| cow manure (without digging under) | 0,3 | 0,5 | 0,17 | 0,35 | 25 |
| cow manure (with digging under) | 0,42 | 0,5 | 0,25 | 0,48 | 25 |
| leather meal | 7,0 | ||||
| present | |||||
| lye water | |||||
| 0,8-1,2 | |||||
| tannery waste | 1,4 | ||||
| 1,3 | 1,2 | 30 | |||
| molasse rinse | 3,0 | 10,5 | |||
| 40 | |||||
| marl | |||||
| 20-90 | |||||
| mussel meal | |||||
| 60 | |||||
| weed (crushed) | 0,5 | 0,7 | 0,2 | 0,2-1,0 | 2 |
| horse manure | 0,44-0,58 | 0,35-0,53 | 0,28-0,36 | 0,21 | 30 |
| apple porridge | 0,26 | 0,24 | 0,1 | 0,04 | 70 |
| debris (sifted) | |||||
| 20-60 | |||||
| reed | 0,6 | 0,26-0,67 | 0,2 | traces | 50 |
| sewage sludge | 0,36 | 0,16 | 0,15 | 2,1 | 19 |
| soot | 3,5 | 1-2 | 0,5 | 4-10 | 90 |
| sheep manure (without digging under) | 0,56 | 0,16 | 0,32 | 0,28 | 32 |
| sheep manure (with digging under) | 0,83 | 0,66 | 0,23 | 0,35 | 32 |
| foamy soil | 0,2-0,5 | ||||
| 0,5-1,5 | 15-30 | 15-30 | |||
| rinse | |||||
| 0,5 | |||||
| stable manure | |||||
| stable manure (3 to 5 months) | 0,55 | 0,65 | 0,3 | 0,73 | 30 |
| street waste | 0,5 | ||||
| 1-10 | present | present | |||
| straw | |||||
| rye | 0,45 | 1,0 | 0,26 | 0,29 | 85 |
| wheat | 0,45 | 0,9 | 0,2 | 0,28 | 85 |
| barley | 0,5 | 1,0 | 0,2 | 0,33 | 85 |
| peas | 1,4 | 0,5 | 0,35 | 1,82 | 80 |
| trimming waste material | traces | 0,74 | 0,3 | present | 20-60 |
| urine | 0,35 | 0,21 | 0,27 | 0,02 | 2 |
| pig manure | 0,45 | 0,61 | 0,19 | 0,08 | 30 |
| meat meal | 5,8 | 0,3 | 17,4 | 22,3 | 40 |
| fly ash | 0,5 | 12,0 | 0,5-1,5 | 15 | traces |
When and how to fertilize
[edit | edit source]The timing: depends on the soil type. Sandy soils are best foreseen of compost or old manure in february. On clayish soils this is best done in autumn. When we would fertilise sandy soils in autumn, a large part would be rinsed to deeper soil layers, where it is unreachable by the plant roots. This is an issue specific to light/sandy soils due to their already loose structure. On clayish soils with a a dense structure, the trough-flushing is low. By already fertilising in autumn, the clods produced by the soil improver can freeze-break in winter.
The digging-in of the manure. In most gardens the manure is simply dug in, this is however not recommended. By simply digging-in, we destroy many of our soil organisms. Each type of micro-organism lives at its own depth in the soil. By digging and turning the soil around, we bring the topsoil a depth where many micro organisms suffocate. A good way to process manure and compost, without disturbing the soil too much is to dig shallowly. It is sufficient to dig in the fertilizer in the 5 to 10 cm-layer under the soilsurface.
Fertilizers
[edit | edit source]Artificial fertilizers
[edit | edit source]Nitrogen: several N-compounds exist:
- Nitric N: works fast, but washes out easily
- Ammoniacal N: works slow (needs to be converted to nitrical first), washes out less easily (locked to clay-humus-complex)
- Urea N: works rather slow (needs to be converted to ammoniacal and then to nitric N first)
- Amidic N: works very slow (needs to be converted to ureum, then to ammoniacal, and then to nitric N first)
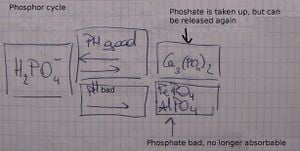
Phosphor: phosphor= directly absorbable by H2PO4- (phosphate)
| Fertiliser | Main constituents | Base equivalent * |
| Nitrogen fertilisers: | ||
| ammonia nitrate 22% N | ½ ammoniacal nitrogen | |
| ammonia nitrate 26% N | ½ nitric nitrogen | |
| ammonia sulphate 21% N | ammoniacal nitrogen | |
| ammonia sulpho nitrate 26% N | ammoniacal + nitric nitrogen | |
| Liquid ammonia 82% N | ammoniacal nitrogen | |
| Calcium nitrate 15,5% N | nitric nitrogen | |
| Chili nitrate 16% N | nitric nitrogen | |
| Kalcyanamide 18% N | amidic nitrogen | |
| Ureum 46% N | ureum nitrogen | |
| Phosphorus fertilisers: | ||
| Superphosphate 18% P2O5 | ||
| Metal slags 15-18% P2O5 | ||
| Rhenaniaphosphate 38% P2O5 | ||
| Triple superphosphate 43% P2O5 | ||
| 0 | ||
| Soft phosphates (minimum 25%) | ||
| Potassium or potash fertilisers: a distinction is made between chlorinated and non-chlorinated potassium/potash fertilisers | ||
| Potassium chloride 40, 50 or 60% K2O | KCl | 0 to +2 |
| Potassium sulphate 48% K2O | K2SO4 | |
| Potassium soda 20% K2O and 28% Na2O | KCl + NaCl | |
| Patented potassium 26% K2O and 8 to 12% MgO | K2SO4 + MgSO4 | |
| Magnesium fertilisers: | ||
| Magnesium sulphate 16% MgO | MgSO4 | |
| Kieserite 27% MgO | MgSO4 | |
| Magnesite 90% MgO | MgO |
For magnesium limes: see lime and soil improvers
- If the base-equivalent is negative, the fertilizer is acidifying
Complex compound fertilizers
[edit | edit source]Complex compound fertilizers contain NPK (Mg) in each pellet. eg: 10-15-20(5) means 10% N, 15% P, 20% K, 5% Mg
Calculation of quantities
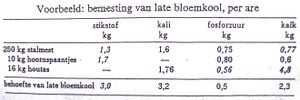
[edit | edit source]Eg: A crop like cauliflower requires 3 kg of N per are 100 kg of ammonia contains 20 kg of N 10 kg of ammonia contains 2 kg of N To obtain 3 kg of N, we thus need to provide 15 kg of ammonia.
Lime fertilizers
[edit | edit source]Workings of lime in the soil
[edit | edit source]- Structure: Ca makes heavy clayish soils less stiff, and improves the coagulation.
- pH: lime fertilizers increase the pH when the soil is too acidic
- Enriching of the soil with N: a reduction of the level of acidity promotes the development of N-binding bacteria, hereby improving the the binding of N
- Mobilization of plant nutrition: the present food supplies are rendered absorbable
At a low pH, the uptake of N, K, Mg, S decreases and the solubility of trace elements is increased with a too acidic pH. At a high pH, the uptake of Fe, Mn, Cu, Zn decreases.
Liming is more focused on improving the soil structure and soil pH then the supplying of crops with Ca.
Lime fertilizers
[edit | edit source]- Slaked agricultural lime powder Ca(OH)2: abv= 50-60
- Calcium carbonate lime such as marl lime, chalc, carbonated agricultural lime CaCO3 abv= 35-52
- Calcium and magnesium carbonates such as dolomitic lime MgCO3 and CaCO3 abv= 45-60
- Magnesium lime powder Mg(OH)2 -Ca(OH)2 abv= 50-60
- Foaming soil CaCO3 abv= 20-40
Acid binding value
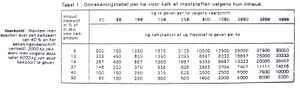
[edit | edit source]In a liming advise, the lime dosage is given in kg abv per ha. Eg: calcium dosage of 1000 kg abv per hectare, with the use of agricultural lime powder with a abv=50 means the administration of 2000 kg/ha.
When to administer
[edit | edit source]Repair liming: When the pH is too low and a lot of lime needs to be administered, it is best done in late summer or autumn, and spread over 2-3 years. The lime needs to be scattered and well mixed with the soil. Maintenance liming: slight limings to supplement the lime store is best done in early spring after plowing and then well mixed.
See also
[edit | edit source]- Agriculture manual 1 2 2
- Sulphur
- Legumes
- Integrated soil fertility
- Soil substrates and fertility
External links
[edit | edit source]| Authors | |
|---|---|
| License | CC-BY-SA-3.0 |
| Cite as | KVDP (2006–2025). "Fertilizers". Appropedia. Retrieved June 4, 2026. |