Hand sanitizersW are becoming ubiquitous in the developed world, commonly found in public places such as shopping malls and waiting rooms. These hand cleaners contain a mixture of alcohol, water and a thickening agent to help the solution spread completely over the hands. Some solutions may contain moisturizers, perfumes or other non-medical additives. Alcohol-based solutions are known to eliminate a significant portion of germs carried on the hands, and are also used in medical facilities to sterilize equipment and surgeons' hands,,.[1][2][3]
This work proposes a method to produce hand sanitizers containing alcohol from local, natural resources by fermenting available sugars and starches from corn or potatoes, and combining the result with a thickening agent to increase spreading of the solution over the area to be cleaned before the alcohol evaporates. A comprehensive review of hand washing techniques and best practices can be found here, as provided by the Ontario Ministry for Health and Long-Term Care.
The target audience for this method are people in the developing world that would benefit from cleaner medical facilities and a reduction in illness and diseases spread by unclean tools. The method of production outlined below has not yet been tested, however it has been verified theoretically to effectively produce ethanol from the glucose contained in the original feed. The scale of production may be such that one person or party can satisfy the needs of their village, or perhaps would be able to sell the product at a market to people from neighbouring villages.
Production from food stuffs will be controversial in regions where food sources are limited to start, however reducing the spread of infection may improve overall health in the village to the point that the benefits outweigh this cost. This is a very difficult question to answer globally, and should be addressed on a regional basis. The best practice may be for a single producer to sell the product at market, although this sort of a monopoly may lead to the charging of exorbitant rates for the compound.
A mind map has been developed to indicate the projected flow for this investigation and will be updated as progress is made. (Mind maps created using MindMeister's free service.)
Introduction[edit | edit source]
A natural hand sanitizer should be composed of materials for production and use in the developing world, with its primary function being to reduce risk of infection during medical procedures such as surgery. In its simplest form, ethanol-based hand sanitizers have two components, each with a specific task and each from naturally occurring sources or compounds:
- Alcohol: Typically either ethanolW or isopropanolW is used in the solution to kill bacteria known to be the primary transmitter of illness and cause of infection. The quality and effectiveness of the hand sanitizer is dependent on the volume-fraction of alcohol contained in the solution, with a study by the Center for Disease Control (CDC) reporting 60% alcohol content to be more effective at eliminating bacteria and fungii from the skin than a 40% solution.[4] A study by Ontario's Provincial Infectious Disease Advisory Council (PIDAC) found ethanol content in the range of 70-90% to be most effective at killing bacteria provided the hands are not visibly soiled (download the report here).[5] This method is deigned to produce ethanol to by distilled in multiple steps to produce sufficiently high concentrations to be effective.
- Thickening Agent: A thickening agent is included to slow the rate of evaporation of the alcohol and increase the 'wetting time' on the surface of the skin. The exact thickening agent used depends on the type of alcohol which governs solubility of all other components. Examples include gelatinW (derived from colagen in the bones and skin of animals, and presenting cultural limitations) and calcium acetateW. The rate of ethanol evaporation is slowed by the addition of the thickening agent,[6] although the exact ratio is dependent on the sources of both ethanol and the compounds used to produce the thickening agent as this 'homemade' recipe may see significant differences in composition between batches.
Both of the above components will be developed through appropriate processes, and alternative sources or methods of production will be provided where possible. Also, if the materials required to produce the thickening agent are not available the ethanol can be used independently, possibly with distribution via spray bottle aiding in increasing coverage before evaporation.
It is the responsibility of the user to ensure adequate coverage of the hands during application, despite the inherent effectiveness of these types of solution. There are several spots on the hand that are frequently missed, so particular care should be taken during application.
Technical Specifications[edit | edit source]
The procedure required for producing ethanol from potatoes is relatively straight forward and most of the processes' time commitment is in allowing the fermentation process to occur. That is, very little time is required from a skilled labourer meaning that little time is taken from other essential activities within the community. Furthermore, the process of fermentationW is well understood and by adding yeastW to a mixture high in glucose and maintained at modest temperatures (approximately 30 degrees C), fermentation is inevitable. A standard biochemical pathway is followed, from which yeast digest glucose ("reactants") and release ethanol and carbon dioxide ("products") in a process known as glycolysisW.
Note: The following procedure has not yet been carried out by the original writer, but has been carefully considered for technical accuracy.
A detailed description of the procedure and equipment is provided here, complete with schematics and estimates of time requirements.
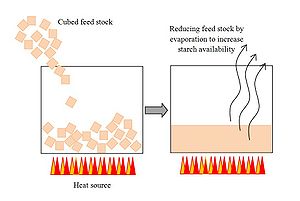
Cut whole, washed potatoes (dirt removed) into small cubes and bring to a steady boil in a pot over a fire or heating element. The smaller the cubes, the more quickly they will cook through. Since the potatoes are not going to be eaten, the goal of the first step is to reduce the mixture to a mush which increases the availability of starches for the yeast in the steps that follow. Also, the boiling action serves to sterilize the potatoes from any contaminants they may bring into the solution. (Note that a particular temperature is not crucial at this stage, and time will vary based on the quantity of potatoes and water used.)
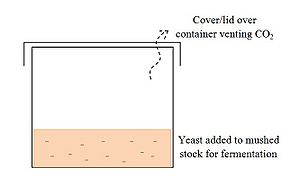
Remove the potato mixture from the heat source, and let it cool to around 30-35 degrees C. Remove a small portion of the slurry to be used to feed the seed stock in the next step. Add yeast to the mixture and let stand for approximately one week in a closed container, occasionally opening the container slightly to vent the CO2 and prevent a build-up of pressure. Alternatively, a hole could be cut in the top of the container and a rubber sheet or balloon fixed to the inside of the lid of the container, pierced with a pin. When the pressure builds up to a level that causes the rubber to expand and open the pinhole enough for CO2 to escape, the pressure will be reduced. This will maintain a pressure equilibrium in the system without requiring opening of the container and further risking contamination.
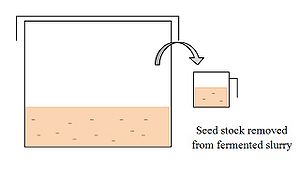
Remove a portion of the fermented slurry (potatoes and yeast) from the fermentation container to seed the next batch for fermentation. This 'seed' batch will contain some of the potato mixture and some of the yeast, which will allow the yeast to then be stored for several days. The semi-active yeast will consume the starchy mix, replicating by budding and replenishing the yeast source in preparation for the next fermentation batch. Add the fresh (unfermented) potato slurry removed in the previous step to the seed stock so that the yeast has some fresh glucose on which to feed and replenish itself.
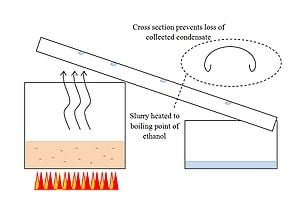
Once fermentation is complete and a seed batch has been removed, it is time to extract the ethanol from the fermented mixture. Since ethanol evaporates at approximately 78 degrees C[7] and water boils at 100 degrees C, this can be accomplished by heating the mixture to some temperature in the range 78-100 degrees C (i.e. 80, or 82 degrees C) where the ethanol will evaporate and can be collected by a condenser but the water will remain in solution. The condensate can then be re-distiled as necessary to achieve sufficiently high alcohol concentration to produce an effective hand sanitizer.
Note that the cross-sectional shape of the condensate collector is such that all of the evaporate that collects on the condenser surface will be saved and directed to the lower container. The small bends on the outer edges of the trough prevent condensate from running down the side of the trough and dripping to the ground.
Production of the thickening agent (calcium acetate) can be accomplished by soaking egg shells in vinegar for extended periods of time (approximately two weeks), then evaporating the vinegar from the solution. One will then be left with a powdered form of calcium acetate which the ethanol can be added to slowly to achieve the desired viscosity for rubbing on the hands. The exact ratio of thickening agent to ethanol (or solution, as 'ethanol' will actually be a solution of 70-90% ethanol and 10-30% water) has yet to be determined, but should be done so experimentally. Also, it is unclear as to whether or not the calcium acetate thickening agent will leave any undesired residue on the hands of the applicant; this will be investigated during testing of the solution.
Engineering Aspects[edit | edit source]
As we see in the procedure, much of the production is carried out by biochemical processes of fermentation that cannot be significantly altered, even in the developed world by trained Chemical Engineers. However, there are a few techniques or tricks that can be employed to minimize losses due to system hardware and to maximize product yield.
- Ensure that the size of feed stock is reduced (by cutting, cubing, shreading, etc.) to decrease rates of reduction of starches, and increase glucose availability for yeast during fermentation.
- Ensuring container is adequately sealed during fermentation to prevent oxygen or other contaminants from entering system and introducing secondary, competing chemical processes that consume the reactant (glucose) and do not produce useful product (ethanol).
- Properly collecting ethanol condensate to ensure none/little is lost during distillation.
- Controlling temperature by control of heat source, such as by the air/fuel ratio of an open flame.
- Proper storage of flammable components during preparation, production and storage of the sanitizer and its components.
- Reducing contaminants to prevent the formation of toxic components or competing side-reactions reduce yield.
Medical Aspects[edit | edit source]
While hand washing is essentially taken for granted in the developed world, it is important to note the strong evidence linking hand washing to reductions in transmitted bacteria and viruses. The following reviews by reputable institutions discuss the benefits of hand washing in general, as well as the specific benefits of an alcohol-based hand disinfectant over traditional soap-and-water techniques. By demonstrating the effectiveness and considering limitations that must be met or exceeded to produce effective solutions, rates of transmitted infection can be reduced in the developing world as they have been in the developed world.
- Mayo Clinic: Hand Hygiene - A frequently missed lifesaving opportunity during patient care (Download 59 kb PDF here)
- Infection Control and Hospital Epidemiology, Chicago Journals: No Time for Handwashing!? Handwashing versus Alcoholic Rub: Can We Afford 100% Compliance? (Download 791 kb PDF here)
- Division of Hospital Epidemiology, University Hospitals, Basel, Switzerland: Replace Hand Washing with Use of a Waterless Alcohol Hand Rub? (Download 85 kb PDF here)
- The origins of hand washing in medical facilities (Download the 343 kb PDF here)
Regional Considerations[edit | edit source]
The process has been developed to consider locally available materials and resources, but does assume access to some more 'advanced' components that may need to be purchased from regional markets. Alternatives to these are being explored, but all efforts have been made to develop a simple, sustainable process.
Since food sources are to be used as feed for the process, regions that already suffer from low availability of such resources may choose not to produce such compounds, by may be able to purchase them from a market on an as-needed basis. It is interesting to note that in recent years, global potato production has shifted from the majority being produced in the developed world to the developing world.[8]
The proposed thickening agent (calcium acetate) was selected for two reasons: (1) it can be produced from local materials at low cost, with little skill and equipment; (2) it does not carry the same sort of cultural limitations that another common thickening agent (gelatinW) does. Gelatin is made from collagen derived from the skin and bones of animals so several cultures and religious groups avoid its use and/or consumption since the source is considered sacred. This is not the case with calcium acetate, allowing for relatively easy and widespread adoption.
Skills and Knowledge[edit | edit source]
Although the procedure seems to be complex, most of the work in producing the ethanol in particular is done by the yeast via biochemical pathways in a process known as ethanol fermentationW. This complex process involves the conversion of glucoseW to pyruvateW (via glycolysisW) which is then anaerobically converted into ethanol and CO2 via fermentationW.
Care needs to be taken to restrict, and the public (potential consumers) of the finished product need to be educated on the dangers of consuming the solution. As methanol may also be produced during fermentation in a competing process (which consumes glucose, the fuel for the desired process) and methanol consumption has been known to cause blindness and/or death in humans, its production should be minimized or avoided. (View the material safety data sheet for methanol here.) However, in the home-distillation industry, there are conflicting views on the subject:
- Some sources[9] claim that by discarding the first 50 mL of a 20 L fermentation batch, the methanol will be discarded from the system as methanol evaporates at approximately 64 degrees C and ethanol evaporates at 78 degrees C. Since methanol has the lower boiling point, it will evaporate first during heating of the solution.[10] Once the methanol has been extracted from the solution, it will not re-enter the solution and can be considered 'removed' and the danger eliminated.
- While some sources comment on ways to remove the methanol, and caution heavily against its consumption due to the health risks associated with it, others argue that only very small quantities of methanol (also known as wood alcohol) are produced from fermentation and can in fact be neglected or ignored,[11] and that instead of causing blindness or death, it may the root of the hangover resulting from alcohol consumption.
In the opinion of the original author of this article, it would be best to err on the side of caution and to discard the first portion of condensate collected during the distillation step in order to reduce the risk of incurring the health affects that are known to accompany methanol ingestion or absorption through the skin.
Estimated Costs[edit | edit source]
Raw Material Costs[edit | edit source]
Note that prices are in Canadian dollars, and will vary significantly based on geography.
- Potatoes (or corn, wheat) - approximately $0.40/lb or $0.89/kg here
- Yeast - $5.19/lb or $11.50/kg here
- Metal containers - Old containers can be reused, as long as they can withstand the heat of cooking (approx 100 degrees C)
- Metal sheet - Can use scrap sheet, as long as it is washed. To be bent for condensate collection, so must be somewhat workable.
- Heat source - Can use current fire source or build new fire burning local materials.
- Shovel to dig hole for temperature 'controlled' storage - Could also fashion a metal or wooden scoop to dig the hole.
- Thermometer for temperature monitoring - To approximate temperature control during distillation and fermentation
Other Resources[edit | edit source]
- Time - Up to two weeks to produce (start to finish) due to fermentation and distillation processes.
- Reallocation of food source - May not be an option in some regions, see Regional Considerations
Mistakes and Precautionary Measures[edit | edit source]
Alcohol Poisoning - Due to the relatively high alcohol content in some commercially available hand sanitizers, reports of young children becoming intoxicated and at risk of alcohol poisoning after ingesting the sanitizing liquid have been published. This fact, combined with the risk of methanol poisoning, contribute to the fact that consumption of the solution should be strongly discouraged due to safety considerations.
CO2 Poisoning - Fermentation is the biochemical process by which glucose (sugar) is converted into ethanol with a known biproduct being carbon dioxide (CO2). As such, care must be taken to adequately vent the space where fermentation will take place despite the process being carried out in sealed containers. Even small CO2 leaks can have negative effects on human health and well-being including drowsiness and death.
Methanol Poisoning - Methanol may also be produced by side reactions and must be extracted. Testing and trial attempts should be made to determine the relative quantities of ethanol and methanol produced by the reaction, and methods of extracting the methanol from solution should be explored. When consumed, methanol can lead to blindness or death, but can also be absorbed through the skin in low concentrations and may have negative effects on human health.
Flammability - Due to high ethanol content of the product and some of the intermediate steps of the process, care must be taken to not expose any ethanol-containing product to a direct flame as it is extremely flammable, especially in its pure state. Properly leak-proof and spill-proof containers should be used to reduce this risk. See this videofor an example of the burning of an ethanol gel.
Burn Risk - Since the ethanol extraction process (distillation) is carried out over a heat source such as a fire, care must be taken around the hot elements of such a system. Containers will also heat up to 70-80 degrees C during the process, introducing the requirement for some sort of glove, sling or rack to remove the hot containers from the heat source. Since the shape of such containers will vary as a function of local resources and availability, a unique solution cannot be proposed, however the implemented solution should be reusable, strong (capable of holding container full of hot, fermented potato mash) and be used by one or two operators depending on the size, shape and weight of the container.
Yeast Reuse and Effectiveness - YeastW are relatively resistant organisms with many operating most efficiently at approximate temperature ranges from 20 degrees C to 40 degrees C. Heating them above this temperature may kill the organisms, while storage below this temperature will essentially render them inactive as they 'hibernate'. During hibernation, yeast still requires a food source (glucose), but will consume it much more slowly, and with their effectiveness decreasing over time. Storing yeast supplies should be done so in a refrigerated environment if possible, and if not the yeast should be given a fresh food source (as in Step 3) such that they remain active and fed sufficiently to sustain life and replenish their numbers through budding.
Conclusions[edit | edit source]
Ethanol can be produced in a relatively safe, inexpensive manner from locally available ingredients. Precautions should be taken to avoid consumption of harmful compounds that may be produced during the process, such as methanol (see debate points, above) and other compounds that may be produced by side- or competing-reactions during the fermentation process if unclean equipment is used. If the process is completed successfully and with care, it can essentially be a self-sustaining process with only a new feed source required for each batch.
See also[edit | edit source]
References[edit | edit source]
- ↑ Infection Control Today, Choosing an Alcohol Hand Sanitizer Online, Accessed: Apr/08/2010. http://www.infectioncontroltoday.com/articles/361feat4.html
- ↑ Hand Hygiene, Are Alcohol-free Hand Sanitizers Better Than Alcohol-based Hand Sanitizers? Online, Accessed: Apr/08/2010. http://web.archive.org/web/20110723142934/http://www.handhygiene.net/antisepsis/alcohol-free-vs-alcohol-based.html
- ↑ New York Times, Hand Sanitiers, Good or Bad? Online, Accessed: Apr/08/2010. http://www.nytimes.com/2006/03/21/health/21cons.html
- ↑ CDC Hand Sanitizer Alert. Online. Accessed Apr/05/2010. http://web.archive.org/web/20110906063853/http://www.cdc.gov:80/ncidod/EID/vol12no03/05-0955.htm
- ↑ Best Practices For Hand Hygiene In All Health Care Settings. PIDAC. Revised January 2009. Online. Accessed Apr/05/2010. http://web.archive.org/web/20100714011722/http://www.health.gov.on.ca:80/english/providers/program/infectious/diseases/best_prac/bp_hh_20080501.pdf
- ↑ Novel evaporation-inhibited alcohol-based deicers/anti-icers Online. Accessed Apr/05/2010. http://www.freepatentsonline.com/EP0490796.html
- ↑ Ethanol Preparation and Manufacture', University College Cork, Ireland. Online, accessed Apr/12/2010. http://web.archive.org/web/20161119122937/http://www.ucc.ie:80/academic/chem/dolchem/html/comp/ethanol.html
- ↑ http://www.potato2008.org/en/world/index.html International Year of the Potato, World potato production.
- ↑ http://web.archive.org/web/20120125061451/http://homedistiller.org:80/methanol.htm Home Distiller, Will I go blind?
- ↑ http://web.archive.org/web/20120104122001/http://homedistiller.org/dtw.htm Home Distiller, Removing the methanol.
- ↑ http://www.home-distilling.com/HD_health.asp Home Distilling, Health and Safety.