Z-Plasty/Thinking Like a Reconstructive Surgeon
| Part of | Z-Plasty V-Y Advancement Flap |
|---|
In order to successfully approach any surgical reconstruction, whether it be for a wound, deficit, or condition originating from burns, trauma, cancer, infections, or congenital conditions, it is beneficial to think through the available reconstructive options using the Reconstructive Ladder or Elevator. This section will discuss this concept as well as the process of applying it.
The Reconstructive Ladder/Elevator
[edit | edit source]History
[edit | edit source]While there are documents to suggest that the concept of a reconstructive or wound closure ladder dates as far back as 2600 BCE to Ancient Egypt1, the modern iterations that are frequently referenced by today's reconstructive surgeons trace back to concepts championed by modern founders of plastic surgery including Sir Harold Gillies, Sir Archibald McIndoe, Stephen Mathes, and Foad Nahai. The basic premise was an organization of the possible options for reconstruction in order of increasing difficulty or complexity to help standardize the thought process behind the reconstructive selection, and, in doing so, identify the "simplest" option that would successfully achieve wound closure.2
However, over time, with an increasing number of reconstructive options available to surgeons, improved technology, and more extensive surgical training resulting in greater technical expertise, this concept had to adapt from its initial simplification of the reconstructive process to adequately incorporate these changes. Two of the most vocal proponents of this change were Gottlieb and Krieger3 who published an article introducing the Reconstructive Elevator. Their premise was that the surgeon should select the most appropriate reconstructive procedure for a patient not solely based on choosing the simplest technique that would achieve wound closure, but rather, the technique that would result in the most optimal reconstruction for the patient, their needs, and the overall clinical context with the least morbidity possible. Instead of progressing from one rung of the ladder to the next, one could freely "take an elevator" to the top or any other level as needed to achieve that optimal reconstruction.
This is where the true mastery and art of surgery comes into play as surgeons must select the right procedure for the right patient at the right time and in the right place. In other words, the selection of which level or option on the reconstructive elevator to go to is dependent upon patient factors (i.e. biology, prognosis, beliefs, social circumstance), healthcare system factors (i.e. available staff, supporting services, existing infrastructure and equipment/supplies), and the overall expected outcome of the reconstruction (i.e. form, function, feeling, and recovery time) for the patient. While there are often many ways that a surgeon could achieve wound closure for a patient, it is only by taking into consideration all those intrinsic and extrinsic factors that the most appropriate reconstruction can be identified for any given individual. With that in mind, the following sections will present the Reconstructive Elevator (Ladder) concept as well as an introductory level discussion of the thought process on how to apply it to clinical scenarios.
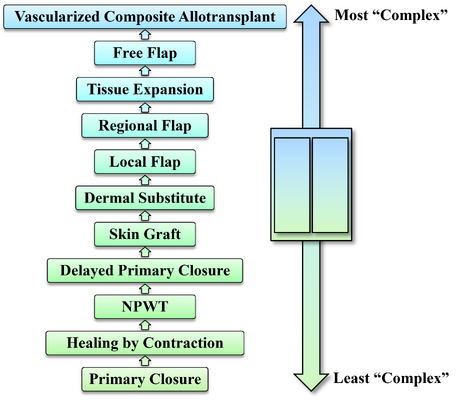
Concept Visualization
[edit | edit source]Reconstructive Elevator (Ladder)
[edit | edit source]The visual representation of the various reconstructive options is classically arranged bottom-to-top from least to most "complex." The implication of complexity is open to interpretation and is often the reason that various orders exist for the different options depending on which surgeon or author is asked. As a result, many of these different permutations have been published and justified in various ways. For the presented version below, the Reconstructive Elevator has been ordered from least to most "complex" with the relative complexity of each level being determined by considering the technical difficulty, the time needed to complete reconstruction, resources required, and extent of post-intervention recovery.
Reconstructive Options: Definitions, Pros, & Cons
[edit | edit source]Primary Closure
[edit | edit source](4,5)
- Definition: Immediate closure of wound during its acute phase using sutures, staples, glue, adhesive strips, or other wound closure device or substrate.
- Pros: Achieves immediate wound closure and often the least amount of wound care, therapy, or other post-intervention requirements compared to other methods)
- Cons: Prematurely closing the wound prior to achieving adequate debridement, microbial control, or demarcation of viable and non-viable tissues can result in wound healing failure, infection, necrosis, excessive dead space, and other sequelae causing suboptimal outcomes and possibly necessitating further, more complex interventions
Healing by Contraction
[edit | edit source](6–8)
- Definition: Performing appropriate wound care through adequate hygiene, debridement, and dressings while the wound naturally contracts and/or re-epithelializes (if hair follicles and other sources of epithelial stem cells are present) to closure from the periphery without assistance from closure devices or other procedures.
- Pros: Does not require surgery, anesthesia, or additional equipment/supplies/expertise. This can most often be done in an outpatient setting with or without skilled assistance and avoids the risks of surgical intervention.
- Cons: Longest time to achieve wound closure with the increased risk of biofilm formation and infection, wound desiccation if wound care is inadequate, hypertrophic scarring, and functionally relevant contractures. Often necessitates frequent follow-up visits (i.e. weekly or monthly) until healed. If it does not heal appropriately, it will eventually need surgical intervention.
Negative Pressure Wound Therapy (NPWT)
[edit | edit source](9–13)
- Definition: Application of negative pressure to the wound using any device that creates a vacuum seal over the entire involved area in order to create a pressure gradient and mechanical forces that promote wound contraction, angiogenesis, and edema reduction.
- Pros: Accelerates wound closure via mechanical contraction, provides some level debridement, decreases microbial burden compared to wound care alone, negates the need for frequent and often painful or inconvenient dressing changes, reduces interstitial edema, and promotes angiogenesis in the wound bed.
- Cons: Requires ongoing access to NPWT device or supplies (most of which require access to electricity for charging, ongoing costs for equipment usage, limited or no access in many countries), may impair mobility due to the size and weight of the device, can be costly to the healthcare system and patient, often requires a skilled wound care specialist to do dressing changes at home or in clinic visits, and may delay wound assessment compared to dressings that are changed daily.
Delayed Primary Closure
[edit | edit source](14,15)
- Definition: performing wound closure through surgery or other wound closure device outside the acute phase of the wound.
- Pros: Allows additional time for wound evaluation and demarcation to ensure sufficient debridement of nonviable tissues and minimizes risk of prematurely closing contaminated or infected wound. Ideal for bites and other forms of trauma that introduce significant microbial and debris burden.
- Cons: Prolongs time to closure with interim period of wound care (and all the challenges that come with it) while increasing risk desiccation of underlying vital structures or prolonged inflammation that contributes to hypertrophic scarring. Requires return to the operating room or procedural room for surgical closure or device application.
Skin Graft
[edit | edit source](16–18)
- Autograft: Harvesting partial or full-thickness skin from a distinct donor site on the patient and grafting it at the site of reconstruction to achieve dermal coverage.
- Pros: Able to achieve rapid coverage of a wound (i.e. often within 1 week) once appropriate wound bed is obtained, can be done with simple instruments, and available donor sites are often many and personalizable to the individual patient.
- Cons: Donor site can be painful and result in permanently hyper- or hypopigmented skin, hypertrophic scars, contractures, and functional deficits. Additional time required to heal donor site as well as for skin graft to adhere and integrate in comparison to primary closure or flaps which have faster healing times. At risk for failure of graft survival due to sheer forces, desiccation, infection, hematoma, malperfusion, and dysthermia. Failure requires harvesting a new skin graft from a different donor site or continuing wound care until the original donor site has healed for repeated harvesting.
- Allograft (homograft) & Xenograft:19–21 Full skin or dermal components obtained from human cadavers or non-human sources (i.e. bovine or porcine).
- Pros: Available in high quantities and can be re-applied as needed. Serves as a temporary coverage option until patient can be stabilized to tolerate a more complex reconstruction or until autologous donor sites are available.
- Cons: Due to immunogenic nature of skin, both homograft and xenograft sources will eventually be rejected by the recipient necessitating additional interventions to achieve ultimate wound closure. Needs to be re-applied when rejected after period of time until definitive coverage options become available. As a result, can be very costly and also requires presence of a tissue bank to store the product. In some cases, repeated use of allograft and xenograft can impact future ability to receive transplants due to histocompatibility issues which are immunogenically created during the rejection process of the foreign skin. In rare instances, viral infections may occur as well as other damaging inflammatory reactions at the grafting site due to rejection processes.
Dermal Substitute
[edit | edit source](19,20)
- Definition: Use of alternative biologically-derived or synthetic substrates meant to replace missing dermis in a temporary or permanent fashion depending on the substitute's properties and method of application.
- Pros: Facilitates rapid coverage of wounds to diminish evaporative and thermal losses as well as their negative sequelae, diminishes pain that often occurs with standard wound care, and can often stabilize wound beds and be used as supporting base for future skin grafting. Can provide additional coverage for implants, titanium plates, and exposed critical structures (i.e. blood vessels, tendons, bone, and nerves) when the flap for coverage is too thin or tenuous.
- Cons: May require additional procedures to remove layers or other components of the substitute as well as apply skin grafts. These substitutes often come at high cost, require storage in a tissue bank or similar facility, and are limited in availability in many countries, which restricts application in low-resource environments.
Local Flap
[edit | edit source](21,22)
- Definition: Mobilizing tissue (e.g. skin, fascia, muscle, etc.) from an autologous donor site adjacent to the site of reconstruction. The various types of flaps and details for flap classification systems will be addressed HERE.
- Pros: Allows for immediate reconstruction of a wound with adaptable, robust, and stable coverage that does not require a distant donor site, complex equipment, or costly substitutes. The local donor often has the best match in terms of skin color, thickness, texture, and hair composition relative to the reconstructed area.
- Cons: Requires adjacent donor site with adequate tissue volume, type, and mobility. Using a donor site adjacent to wounds originating from trauma, infection, and burns may increase risk for failure of these flaps due to unforeseen compromise of these donor sites. An experienced understanding of preoperative planning and markings is needed in order to predict adequate mobilization of the flap(s) and assure vascularity through the entire flap, which increases complexity and training requirements for success.
Regional Flap
[edit | edit source](21,23–25)
- Definition: Mobilizing tissue from a regional donor site that is not adjacent to the reconstructive site and often requires tunneling, islandizing, or pedicle dissection to achieve adequate mobilization to reach the reconstructive site.
- Pros: Increases the number of available reconstructive options and potential tissue types, particularly, those outside the zone of injury, which minimizes unforeseen compromise that can occur in local flaps due to these underlying factors.
- Cons: Techniques required to achieve sufficient mobilization (tunneling, islandizing, pedicaling, perforator dissection, etc.) have increased risk of complications and require a higher level of technical skill. Relying on a distant site may also force the patient to remain in splints or other motion-limiting dressings that impairs mobility and independence in the recovery and rehabilitation period. Some methods may also require a secondary procedure to divide pedicles or modify the flaps.
Tissue Expansion
[edit | edit source](26,27)
- Definition: Implanting an expandable device that is slowly inflated with air or saline over an extended period of time to achieve expansion of overlying tissues.
- Pros: Can be used to expand fasciocutaneous and musculocutaneous flaps so that a greater surface area is available in the flap to achieve coverage using local, well-matched tissue types. May circumvent need for free flap or other more challenging distant donor site reconstructions while also increasing success of local flap reconstruction due to increased vascularity that develops within the flap during the expansion process.
- Cons: Prolongs reconstructive period due to expansion period which involves temporary disfigurement due to the expansion device, discomfort from the expansion process and its impact on mobility, need for multiple and frequent expansions either in clinic or at home, and secondary surgical requirement for expander removal and flap elevation/mobilization to achieve final wound coverage.
Free Flap
[edit | edit source](28–30)
- Definition: Transfer of tissue from donor site to recipient site with defined donor blood vessels that are taken with the flap of tissue and re-connected under microscopic visualization to recipient vessels at the reconstruction site.
- Pros: Provides the greatest flexibility of any autologous reconstructive option as it relates to available donor sites, potential tissue types, size of coverage, complexity of design, and robustness of coverage.
- Cons: Most technically challenging autologous option that requires highly-trained microsurgeons. Often requires long operating times that adds to anesthesia risk for patient, significant resource utilization (i.e. staff, OR time, supplies, etc.), and need for specialized equipment (i.e. surgical microscope, loupes, etc.). Post-operative recovery requires more frequent monitoring from the surgeon, specialized nursing care, extended periods of immobilization, and a generally prolonged recovery time compared to less complex reconstructive methods. Due to the risk of vascular complications during the first 24-48 hours that may require take back to the operating room, the hospital or medical facility supporting staff, systems, and infrastructure at the ready 24 hours per day every day of the week.
Vascularized Composite Allotransplant (VCA)
[edit | edit source](31–37)
- Definition: Transplantation of complex composite tissue units (i.e. face, hands, penis) from matched donor to individual requiring reconstruction.
- Pros: Currently the most advanced reconstructive option for complex wounds and defects given ability to transfer complex anatomic structures (i.e. face or hand) that are not available from autologous, synthetic, or bio-engineered sources.
- Cons: The most technically challenging of all currently available reconstructive options requiring multiple, highly-trained teams to perform simultaneous donor harvesting and recipient preparation and transplantation. Complex coordination of two operating rooms, two teams, and extensive supporting infrastructure central to success. Requires histocompatibility matching between donor and recipient as well as life-long immunosuppression to prevent rejection of the transplanted tissues. Patient selection for VCAs is extremely challenging and complex requiring many different evaluations and considerations related to patient eligibility, psychological readiness, prognosis, social circumstance and support network, post-operative access to care, and ethics; all of which necessitate a large team of specifically-trained surgeons, psychiatrists, immunologists, internists, social workers and case managers, physical and occupational therapists, and nurses.
Future Reconstructive Options
[edit | edit source]Two additional levels that will become significant contributions to the armamentarium of reconstructive surgeons over the next few decades are robotics38–42 and bio-engineered tissues.19,43–45 As technology advances, the complexity of robotic prosthetics with the capacity for fine-tuned haptic feedback and variable force generation will allow for precise, functional, and dynamic prosthetics that may circumvent the need for VCAs and other complex reconstructions that carry additional risks due to immunosuppression, anesthesia time, surgical stress, and donor site morbidity. While there are robotic prosthesis that integrated myoelectric and neuroelectric control mechanisms through implanted electrodes to control mechanical arms and legs, there are still significant barriers related to the interface between machine and biology, specifically the gliosis or scarring that occurs are the electrodes of the control chips. This process of encapsulation of the foreign material results in the eventual loss of conductivity across the interface and need for re-implantation or loss of implant targets altogether. In addition, further advancements in neural coding and computer integrated network are required to alter the sensitivity, feedback, and variable adjustability of the output from these prostheses.
Ultimately, the development of advanced bio-engineering capabilities to create organs and other complex structures in vitro for fully restorative reconstruction through surgical implantation is the future that many surgeons, engineers, and biologists are striving to accomplish. There have been promising studies with simpler structures such as cartilaginous joint surfaces, nerves, and skin sans adnexa; but cost, time, three-dimensional construction, and scaling remain significant challenges for these endeavors. In the future, surgeons will be able to choose between or combine robotic and bio-engineered structures to achieve optimal reconstruction. In the interim, utilization of the aforementioned options currently available to reconstructive surgeons can result in significant restoration of form, function, and feeling.
Applying the Reconstructive Elevator
[edit | edit source]Step 1: Perform Wound Analysis
[edit | edit source]- What tissue types are missing?
- Skin (partial or full thickness, glabrous or non-glabrous)
- Mucosa
- Subcutaneous tissue
- Tendon & Ligaments
- Nerve
- Blood vessels
- Fascia
- Muscle
- Bone
- Cartilage
Reasoning: Achieving optimal reconstruction often requires "replacing like with like" or identifying missing tissues and selecting reconstructions that can restore as much as possible within reason of the overall clinical picture. Beyond the tissue types involved, it is critical to know how much of each tissue type is missing in order to help determine which reconstructive option will provide sufficient volume. It is also important to identify what is remaining and adjacent to the reconstructive site since these remaining tissues may be available as a local donor for use in reconstruction. An important point is that this assessment should be performed after all necrotic or nonviable tissue has been debrided, any foreign debris or traumatic tattooing is removed, and the wound is free from microbial contamination with good source control. Ensuring the wound is adequately debrided and stabilized for reconstruction prior to selecting any reconstructive choice is critical because this preparation often requires the removal of additional tissue, which creates a larger defect and may also impact additional tissue types that may subsequently need reconstruction.
- What anatomic boundaries, relationships, and structures are deformed or absent?
- Anatomic units & sub-units
- Anatomic landmarks
- Anatomic boundaries
- Anatomic relationships/orientation
Reasoning: Not only is it important to know what tissue types are missing, but also their anatomical relationships to one another, the natural landmarks and anatomic units that they create, and the way that they integrate with one another to achieve their original anatomic composition.
- What functions are lost or diminished?
Reasoning: Once the tissue deficits and their structural relations are recognized, it is important to consider the function(s) that this anatomic unit(s) normally perform. For example, is the deficit involving a hand and specific functions that it usually performs? Does the reconstruction need to facilitate pinch, grasp, power grab, or other specific functions? Considering what functions are missing and ideally need to be restored is just as critical as knowing which tissue types need to be replaced and in what relationship to the others.
- What is the chronicity of the wound? (i.e. acute, sub-acute, chronic, or congenital)
- Acute wounds (e.g. abrasion, laceration, incision, burn, blast injury, road rash, or crushing injury)
- Sub-acute wounds (i.e. acute wounds that fail to heal due to infection, patient's underlying conditions, or inadequate treatment but still within the initial healing phase)
- Chronic wounds (e.g. any sort of wound arising due to underlying chronic conditions such as diabetes, vascular disease, chronic pressure, neuropathy or acute wounds that failed to heal and converted to chronic wounds), and can be further classified as:
- "Black" Chronic wounds: those with abundant necrotic and/or infected tissue where the goal is to decrease bacterial load and removal of non-viable tissue usually with thorough mechanical debridement.
- "Yellow" Chronic wounds: wounds with no evident infection with fibrinous exudate and fibrinoid accumulation. The goal is to remove fibrinoid tissue, and is usually achieved by chemical or autolytic debridement if mechanical option not available or viable.
- "Red" Chronic wound: those with granulation tissue (excessive, hypertrophied vascular tissue) needing debridement of colonized granulation tissue – often with a biofilm – and additional procedures (e.g. skin grafts or flaps) to facilitate secondary closure.
- Congenital Conditions (e.g. clefts, craniofacial conditions, meningocele, myelomeningocele, limb malformations, thoracoabdominal defects, etc.)
Reasoning: The "age" of a wound has a direct impact on numerous intrinsic wound factors (e.g. inflammatory state, bacterial and fungal colonization, biofilm burden, fibrosis/fibrinous deposition, granulation, underlying atrophy, and hypertrophic scarring) which must be considered to help guide sequence of reconstruction. As time progress and acute wounds transition to a chronic state, the impact of these temporally-dependent changes become increasingly important in addressing in order to achieve successful reconstruction. With respect to congenital conditions, these are unique pathologies that not only result in deficits of certain tissue types but alterations in the remaining anatomy surrounding the involved areas. Knowing the distinct pathologies involved in a given congenital condition are critical to understanding the reconstructive needs.
Step 2: Identify Patient Goals & Prognosis
[edit | edit source]Reasoning: The wound or defect requiring reconstruction is not an isolated entity but a component of the patient as a whole. Patients often have goals regarding their recovery and expectations for their post-reconstructive functional state that must be considered when determining the best reconstructive option. What specific activities, hobbies, or jobs does that patient want to be able to perform? Are there family, social, and religious beliefs that are helping guide those reconstructive goals for the patient and how can they be integrated into the decision-making process? In addition, the patient's underlying medical conditions, intrinsic biological factors, active concomitant health problems, and overall prognosis related to acute medical state as well as long-term state must be considered. While a patient may desire a certain reconstructive outcome, if they are critically ill or the extent of comorbid conditions significantly increases the risk for perioperative complications then certain options that would otherwise achieve the reconstructive goals must be forfeited for the patient's overall health and well-being.
Step 3: Consider External Factors
[edit | edit source]- Are the necessary surgeons, anesthesiologists, nurses, therapists, and other healthcare providers who are required to perform a given reconstructive option available? (Staff)
- Are the proper supplies, equipment, medications, machines, and other items necessary for the given reconstructive option available? (Stuff)
- Is there sufficient operating room, hospital, clinic, or other healthcare space available in which the specific type of reconstructive care can be provided? (Space)
- Are the necessary supporting systems (i.e. supply chain, referral network, follow-up system, provider communication networks, etc) in place to sustain the chosen reconstructive process? (Systems)
Reasoning: Identifying the necessary staff, stuff, space, and systems that are required to successfully perform each level of reconstruction also needs to be considered. This "Four S" model, originally championed by Paul Farmer, helps providers systematically assess the core components needed for the pre-, peri-, and post-operative care of the patient – which without there cannot be a successful reconstruction. Being able to successfully perform a given reconstruction requires far more than a willing and capable surgeon, so choosing an option that is also realistic and compatible with the healthcare system and environment is critical.
Step 4: Synthesize the Data
[edit | edit source]After proceeding through Steps 1-3, assimilate the understanding of impacted tissue types, anatomic structures/landmarks, and function with the individual patient scenario and the overall healthcare environment to determine which level of reconstruction on the Reconstructive Elevator is most appropriate. Keeping in mind that no wound, patient, or procedure exists in a silo, the interplay of all intrinsic and extrinsic factors must be considered in order to determine the right option for the right patient at the right time in the right place.
Self assessment
[edit | edit source]- Review the contents of this page as well as the rest of the pages related to the Z-Plasty Training Module then go to the AmoSmile App to take the Self-Assessment quiz on the Reconstructive Ladder/Elevator.
- See Z-Plasty Module navigation page for app download instructions.
References
[edit | edit source]- Mazzola RF, Mazzola IC. History of reconstructive and aesthetic surgery. Plast Surg 3rd Ed Lond Elsevier. Published online 2013:11-29.
- Janis JE, Kwon RK, Attinger CE. The New Reconstructive Ladder: Modifications to the Traditional Model: Plast Reconstr Surg. 2011;127:205S-212S. doi:10.1097/PRS.0b013e318201271c
- Gottlieb LJMD, Krieger LMMBA. From the Reconstructive Ladder to the Reconstructive Elevator. [Editorial]. Plast Reconstr Surg. 1994;93(7).
- Eardley WGP, Stewart MPM. Early Management of Ballistic Hand Trauma. J Am Acad Orthop Surg. 2010;18(2):118-126.
- Ogawa R. Surgery for scar revision and reduction: from primary closure to flap surgery. Burns Trauma. 2019;7:7. doi:10.1186/s41038-019-0144-5
- Humphries LS, Kueberuwa E, Beederman M, Gottlieb LJ. Wide excision and healing by secondary intent for the surgical treatment of hidradenitis suppurativa: A single-center experience. J Plast Reconstr Aesthet Surg. 2016;69(4):554-566. doi:10.1016/j.bjps.2015.12.004
- Ovadja ZN, Bartelink SAW, Hadi K, van de Kar AL, van der Horst CMAM, Lapid O. A Multicenter Analysis of Wide Excision and Reconstruction for Severe Anogenital Hidradenitis Suppurativa. Ann Plast Surg. 2021;86(2):193-200. doi:10.1097/SAP.0000000000002554
- Ovadja ZN, Bartelink SAW, van de Kar AL, van der Horst CMAM, Lapid O. A Multicenter Comparison of Reconstruction Strategies after Wide Excision for Severe Axillary Hidradenitis Suppurativa. Plast Reconstr Surg-Glob Open. 2019;7(8):e2361. doi:10.1097/GOX.0000000000002361
- Huang C, Leavitt T, Bayer LR, Orgill DP. Effect of negative pressure wound therapy on wound healing. Curr Probl Surg. 2014;51(7):301-331. doi:10.1067/j.cpsurg.2014.04.001
- Timmers MS, Le Cessie S, Banwell P, Jukema GN. The effects of varying degrees of pressure delivered by negative-pressure wound therapy on skin perfusion. Ann Plast Surg. 2005;55(6):665-671. doi:10.1097/01.sap.0000187182.90907.3d
- Kim P, Attinger CE, Orgill D, et al. Complex Lower Extremity Wound in the Complex Host: Results From a Multicenter Registry. Plast Reconstr Surg-Glob Open. 2019;7(4):e2129. doi:10.1097/GOX.0000000000002129
- Diehm YF, Fischer S, Wirth GA, et al. Management of Acute and Traumatic Wounds With Negative-Pressure Wound Therapy With Instillation and Dwell Time. Plast Reconstr Surg. 2021;147(1S-1):43S-53S. doi:10.1097/PRS.0000000000007610
- Seidel D, Diedrich S, Herrle F, et al. Negative Pressure Wound Therapy vs Conventional Wound Treatment in Subcutaneous Abdominal Wound Healing Impairment The SAWHI Randomized Clinical Trial. Jama Surg. 2020;155(6):469-478. doi:10.1001/jamasurg.2020.0414
- Katiyar AK, Agarwal H, Priyadarshini P, et al. Primary vs delayed primary closure in patients undergoing lower limb amputation following trauma: A randomised control study. Int Wound J. 2020;17(2):419-428. doi:10.1111/iwj.13288
- Rademacher E, Miller PE, Jordan E, et al. Management of Fasciotomy Incisions After Acute Compartment Syndrome: Is Delayed Primary Closure More Feasible in Children Compared With Adults? J Pediatr Orthop. 2020;40(4):E300-E305. doi:10.1097/BPO.0000000000001492
- Knapik A, Hegland N, Calcagni M, et al. Metalloproteinases facilitate connection of wound bed vessels to pre-existing skin graft vasculature. Microvasc Res. 2012;84(1):16-23. doi:10.1016/j.mvr.2012.04.001
- Frueh FS, Sanchez-Macedo N, Calcagni M, Giovanoli P, Lindenblatt N. The Crucial Role of Vascularization and Lymphangiogenesis in Skin Reconstruction. Eur Surg Res. 2018;59(3-4):242-254. doi:10.1159/000492413
- Kalsi R, Messner F, Brandacher G. Skin xenotransplantation: technological advances and future directions. Curr Opin Organ Transplant. 2020;25(5):464-476. doi:10.1097/MOT.0000000000000798
- Shevchenko RV, James SL, James SE. A review of tissue-engineered skin bioconstructs available for skin reconstruction. J R Soc Interface. 2010;7(43):229-258. doi:10.1098/rsif.2009.0403
- Tufaro AP, Buck DW, Fischer AC. The use of artificial dermis in the reconstruction of oncologic surgical defects. Plast Reconstr Surg. 2007;120(3):638-646. doi:10.1097/01.prs.0000270298.68331.8a
- Sabino JM, Slater J, Valerio IL. Plastic Surgery Challenges in War Wounded I: Flap-Based Extremity Reconstruction. Adv Wound Care. 2016;5(9):403-411. doi:10.1089/wound.2015.0656
- Maciel-Miranda A, Morris SF, Hallock GG. Local Flaps, Including Pedicled Perforator Flaps: Anatomy, Technique, and Applications. Plast Reconstr Surg. 2013;131(6):896E-911E. doi:10.1097/PRS.0b013e31828bd89f
- Lu L, Liu A, Zhu L, Zhang J, Zhu X, Jiang H. Cross-Leg Flaps: Our Preferred Alternative to Free Flaps in the Treatment of Complex Traumatic Lower Extremity Wounds. J Am Coll Surg. 2013;217(3):461-471. doi:10.1016/j.jamcollsurg.2013.03.029
- Miller EA, Friedrich J. Soft Tissue Coverage of the Hand and Upper Extremity: The Reconstructive Elevator. J Hand Surg-Am Vol. 2016;41(7):782-792. doi:10.1016/j.jhsa.2016.04.020
- Ullmann Y, Fodor L, Ramon Y, Soudry M, Lerner L. The revised "reconstructive ladder" and its applications for high-energy injuries to the extremities. Ann Plast Surg. 2006;56(4):401-405. doi:10.1097/01.sap.0000201552.81612.68
- Langdell HC, Taskindoust M, Levites HA, et al. Systematic Review of Tissue Expansion: Utilization in Non-breast Applications. Plast Reconstr Surg-Glob Open. 2021;9(1):e3378. doi:10.1097/GOX.0000000000003378
- Knobloch K, Vogt PM. The reconstructive sequence in the 21st century. Chirurg. 2010;81(5):441-446. doi:10.1007/s00104-010-1917-3
- Lineaweaver WC. Microsurgery and the reconstructive ladder. Microsurgery. 2005;25(3):185-186. doi:10.1002/micr.20125
- Levin LS. From Autotransplantation to Allotransplantation: A Perspective on the Future of Reconstructive Microsurgery. J Reconstr Microsurg. 2018;34(9):681-682. doi:10.1055/s-0038-1639367
- Ninkovic M, Voigt S, Dornseifer U, Lorenz S, Ninkovic M. Microsurgical Advances in Extremity Salvage. Clin Plast Surg. 2012;39(4):491-+. doi:10.1016/j.cps.2012.08.003
- Mills E, Felsenheld JH, Berman ZP, et al. Guiding Strategies for the Future of Vascularized Composite Allotransplantation: A Systematic Review of Organ Donation Campaigns. Plast Reconstr Surg. 2020;146(4):922-934. doi:10.1097/PRS.0000000000007187
- Rifkin WJ, David JA, Plana NM, et al. Achievements and Challenges in Facial Transplantation. Ann Surg. 2018;268(2):260-270. doi:10.1097/SLA.0000000000002723
- Caplan AL, Parent B, Kahn J, et al. Emerging Ethical Challenges Raised by the Evolution of Vascularized Composite Allotransplantation. Transplantation. 2019;103(6):1240-1246. doi:10.1097/TP.0000000000002478
- Gray KM, Peterson JM, Padilla PL, et al. Vascularized Composite Allotransplantation in Burn Reconstruction: Systematic Review and Meta-analysis. J Burn Care Res Off Publ Am Burn Assoc. 2021;42(3):465-472. doi:10.1093/jbcr/iraa188
- Shokri T, Saadi R, Wang W, Reddy L, Ducic Y. Facial Transplantation: Complications, Outcomes, and Long-Term Management Strategies. Semin Plast Surg. 2020;34(4):245-253. doi:10.1055/s-0040-1721760
- Safi A-F, Kauke M, Nelms L, et al. Local immunosuppression in vascularized composite allotransplantation (VCA): A systematic review. J Plast Reconstr Aesthetic Surg JPRAS. 2021;74(2):327-335. doi:10.1016/j.bjps.2020.10.003
- Lupon E, Lellouch AG, Acun A, et al. Engineering Vascularized Composite Allografts Using Natural Scaffolds: A Systematic Review. Tissue Eng Part B Rev. Published online July 9, 2021. doi:10.1089/ten.TEB.2021.0102
- Aman M, Sporer ME, Gstoettner C, et al. Bionic hand as artificial organ: Current status and future perspectives. Artif Organs. 2019;43(2):109-118. doi:10.1111/aor.13422
- Farina D, Vujaklija I, Brånemark R, et al. Toward higher-performance bionic limbs for wider clinical use. Nat Biomed Eng. Published online May 31, 2021. doi:10.1038/s41551-021-00732-x
- Sanz-Morère CB, Martini E, Meoni B, et al. Robot-mediated overground gait training for transfemoral amputees with a powered bilateral hip orthosis: a pilot study. J NeuroEngineering Rehabil. 2021;18:111. doi:10.1186/s12984-021-00902-7
- Valle G, Saliji A, Fogle E, Cimolato A, Petrini FM, Raspopovic S. Mechanisms of neuro-robotic prosthesis operation in leg amputees. Sci Adv. 2021;7(17):eabd8354. doi:10.1126/sciadv.abd8354
- Fleming A, Stafford N, Huang S, Hu X, Ferris DP, Huang H (Helen). Myoelectric control of robotic lower limb prostheses: a review of electromyography interfaces, control paradigms, challenges and future directions. J Neural Eng. Published online 2021. doi:10.1088/1741-2552/ac1176
- Lee OJ, Ju HW, Kim JH, et al. Development of Artificial Dermis Using 3D Electrospun Silk Fibroin Nanofiber Matrix. J Biomed Nanotechnol. 2014;10(7):1294-1303. doi:10.1166/jbn.2014.1818
- Wisser D, Steffes J. Skin replacement with a collagen based dermal substitute, autologous keratinocytes and fibroblasts in burn trauma. Burns. 2003;29(4):375-380. doi:10.1016/S0305-4179(03)00013-5
- Parenteau-Bareil R, Gauvin R, Cliche S, Gariepy C, Germain L, Berthod F. Comparative study of bovine, porcine and avian collagens for the production of a tissue engineered dermis. Acta Biomater. 2011;7(10):3757-3765. doi:10.1016/j.actbio.2011.06.020
| Authors | AmoSmile |
|---|---|
| License | CC-BY-SA-4.0 |
| Organizations | AmoSmile, Operation Smile |
| Cite as | AmoSmile (2021–2025). "Z-Plasty/Thinking Like a Reconstructive Surgeon". Appropedia. Retrieved June 4, 2026. |