Bioconversion of Organic Residues for Rural Communities (UNU, 1979, 178 p.)
Indian experience with algal ponds[edit | edit source]
G.S. Venkataraman
Division of Microbiology, Indian Agricultural Research Institute, New Delhi, India
Introduction[edit | edit source]
As early as 1949, Spoehr and Milner (1) suggested that mass culture of algae would help to overcome global protein shortages. The basis for their optimism was that algae had a crude protein content in excess of 50 per cent and a biomass productivity of the order of 25 tons/ha/year. Ironically, in spite of the lamentably low per capita protein supplies in many parts of the world, mass cultivation of algae has received only casual interest. The United Nations Environmental Programme (UNEP) is emphasizing nitrogen fixation and nutrient recycling through a programme that will establish microbiological centres (MIRCENS), and it is hoped that this will stimulate interest in micro-algae technology as a component of an integrated recycling system for rural communities.
There are a number of relatively large production systems for micro-algae, using completely synthetic nutrients. These systems are expensive, not only because of the high cost of land and the technology required but also in terms of water use and the price of inorganic nutrients. Even an optimistic cost extrapolation indicates that production expenses will be US$2 - 3 per pound of crude algae, or US$4 - 6 per pound of crude protein. For reasons of economy alone, it is therefore necessary to use organic wastes for industrialized production of algal protein feed.
In India, we are developing an integrated waste recycling system, in which algal production forms an integral part (2). This is significant from both the biological and the environmental point of view. This system can provide, simultaneously, fuel (methane), feed algae (for fish, livestock), fish, manure for crops, and water for irrigation. The two main objectives of the All India Co-ordinated Project on Algae are: a. to develop rural-based recycling systems involving agriculture-aquaculture-pisciculture, and b. to develop viable, economical, rural-oriented systems for algal biofertilizers for rice that will help to relieve the pressure on chemical nitrogen supplies without impairing crop productivity. This will provide a cyclic instead of a linear system of nutrient supply.
Cultivation of algae in wastes for feed[edit | edit source]
While Spirulina platensis is grown at Delhi (Indian Agricultural Research Institute) and Nagpur (National Environmental Engineering Research Institute), Chlorella is being grown at Pondicherry (Auroville Centre for Environmental Studies). Spirulina, besides its rapid growth rate, high protein content, and lack of a thick cell wall (3), is amenable to simple filtration, giving it an economic advantage over such algae as Scenedesmus and Chlorella.
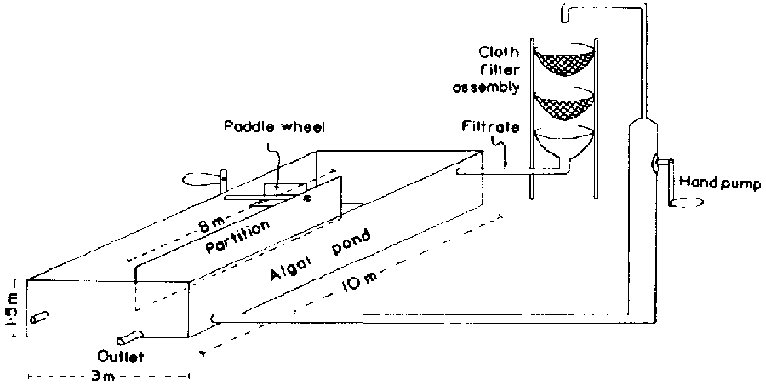
At Delhi, the algal production unit is a 30 m² cement tank with a partition in the middle to facilitate circulation of the algal suspension by means of a hand-operated paddle wheel turned for 30 minutes twice a day (Figure 1). For harvesting, the algal suspension is pumped out by a hand-pump onto a series of cloth filters fitted to wire mesh baskets suspended in a frame. The filtered algal slurry is scooped out of the cloth and sun-dried. The filtrate is then recycled into the production unit. The average yield of algae amounts to about 15 - 20 g/day/m².
Figure. 1. Schematic Diagram of the Spirulina Production Unit at the Indian Agricultural Research Institute, New Delhi
Tables 1 and 2 show the growth potential of Spirulina in the digested slurry effluent from the cow dung gas plant and in cattle urine, respectively, with and without bicarbonate fortification. The slurry effluent supported algal growth at all dilutions even in the absence of added bicarbonate, although addition of bicarbonate (18 9 NaHCO3/l) stimulated algal growth to the level of algae grown in synthetic inorganic nutrient medium. In contrast, pure cattle urine failed to support algal growth in the absence of bicarbonate, presumably because the urine lacks an available carbon source. Supplementation of cattle urine with bicarbonate supported the growth of the algae up to a level of 3 per cent urine, beyond which the urine per se seemed to inhibit algal growth even with addition of bicarbonate.
TABLE 1. Growth Potential of Spirulina platensis in Digested Cow Dung Slurry Effluent, with and without Added Bicarbonate (18 9 NaHCO3/l)
|
concentration of slurry effluent |
pH |
Solids (g/l) |
Nitrogen (mg/l) |
Dry wt. alga (g/l) |
| 1% | 8.7 | 0.23 | ||
| 0.0684 | 25 | |||
| 1% + bicarbonate | 9.2 | 0.63 | ||
| 2% | 8.7 | 0.27 | ||
| 0 1368 | 50 | |||
| 2% + bicarbonate | 9.3 | 0.74 | ||
| 3% | 8.7 | 0.33 | ||
| 0.2052 | 75 | |||
| 3% + bicarbonate | 9.1 | 0.87 | ||
| 5% | 8.7 | 0.32 | ||
| 0.3420 | 125 | |||
| 5% + bicarbonate | 9.1 | 1.08 | ||
| 7% | 8.7 | 0.38 | ||
| 0.4788 | 175 | |||
| 7% + bicarbonate | 9.2 | 0.84 | ||
| 10% | 8.7 | 0.37 | ||
| 0.684 | 250 | |||
| 10% + bicarbonate | 9.1 | 0.94 | ||
| Control (synthetic medium) | 9.2 | - | 400 | 0.9 |
Source: Rao and Venkataraman, unpublished data.
TABLE 2. Growth Potential of Spirulina platensis in Cattle Urine with and without Added Bicarbonate (18 9 NaHCO3/l)
| Concentration of urine | pH | Dry wt. Alga(g/l) |
| 1% | 7.9 | - |
| 1% + bicarbonate | 8.8 | 1.33 |
| 3% | 8.6 | - |
| 3% + bicarbonate | 8.8 | 0.8 |
| 5% | 8.7 | - |
| 5% + bicarbonate | 8.8 | - |
| 7% | 8.7 | - |
| 7% + bicarbonate | 8.8 | - |
| Control (synthetic medium) | 9.2 | 0.91 |
Source: Rao and Venkataraman, unpublished data.
Spirulina has a 50 - 60 per cent protein content with a well balanced amino acid pattern except for a deficiency of sulphur amino acids. The PER is higher than that in Chlorella and Scenedesmus (4) (Table 3). The BV, TD*, and NPU values are 68, 75.5, and 52.7, respectively. Cereals like rice, wheat, and ragi fortified with the alga were used in our experimental diet. Because lysine content is higher in the alga (4.34 9/16 9 N) than in the cereals, an increasing proportion of algal protein in the supplemented diets progressively improved the PER. The best growth pattern was obtained in diets containing alga and rice, each of which contributed 50 per cent of the protein.
TABLE 3. PER, NPU, and BV Values of Different Micro-algae.
| PER | NPU | BV | |
| Spirulina maxim* | 2.30 | 45.6 - 49.8 | 60 - 65 |
| Spirulina platensis | 2.07 | 52 7 | 68 |
| Scenedesmus acutus** | 1.27 | 52 | 72.1 |
| Chlorella ellipsoidea | 0.94 | - | - |
* Clement and Van Landeghem (3)
** Becker et al. (4)
Two types of integrated recycling systems are being developed at the Nagpur centre. In one, domestic sewage is utilized to produce an alga which is then used for fish culture. To adapt Spirulina to raw and settled sewage, a system has been developed in which the alga is initially grown in a synthetic medium that is progressively diluted at regular intervals with raw-plus-settled sewage, and finally with raw sewage alone. By this means, a population of alga that grow profusely in raw sewage has been selected. As sufficient phosphate is present in sewage, no phosphate fortification has been found necessary. However, 2 - 3 g NO3/l are required under such conditions for optimum growth of the alga. In contrast to a high requirement for bicarbonate (18 9 NaHCO3/l) by the alga growing in the synthetic nutrient medium, only a little bicarbonate (2 - 4 9 NaHCO3/l) is required in the sewage medium.
The other approach involves an integrated system of a night soil gas plant, algal culture, and pisciculture. The digesters have a capacity of about 18 m³ and yield about 25 m³ gas per day. The volatile solids loading is kept at about 2.5 kg/m3 /day. Destruction of volatile solids varies from 40 to 50 per cent. The oxidation pond (36' x 18'x 4') is made of earthen embankments with inlet and outlet structures and normally holds about 25 m³ (825 cubic fee feet) of effluent. After digestion, the sludge filtrate is added to the pond. The biological oxygen demand (BOD), chemical oxygen demand (COD), suspended solids, pH, and alkalinity are being studied to arrive at the optimum load for a pond of this size. The efficiency with which this system kills helminths and other parasites will also be determined. The sludge is dried on special drying beds and carted off as manure.
At Pondicherry, the major emphasis is on Chlorella production and harvesting. This requires aeration and rain water collection, the latter achieved by pumping and water delivery systems. The circular algal production unit of about 200 m² illuminated surface directs the circulating culture alternatively in a thin moving sheet and in cool, deeper sections to optimize utilization of light and CO2 as well as to control temperature. Three pre-existing concrete slopes have been joined to the pond to give a total of more than 100 m² to be used for the moving sheet illumination and aeration.
Four venturi devices have been installed in one compartment of the underground reservoir to increase aeration in the circulation pattern. The suction produced evacuates the CO2enriched air from a system of three inter-connected fermentation tanks of 25,0001 total capacity as well as from two composting chambers (each 5 m x 0.75 m x 1 m). The fermentation tanks are heated by the composting chambers as well as by the solar collectors that form the top of the tanks. The complete circuit for the circulation of the culture thus includes an aeration chamber drawing warm, CO2-enriched air from the fermentation tanks, a dark retention period in the underground reservoir, passing through the pump into a thin, rapidly moving sheet over the sloping roof, a distribution channel jetting into a cool, growing ring of 20,0001 capacity that overflows into a thin sheet and collects in the cool central growing basin before again passing into the venturi aerators by gravity.
This circulation can be timed at various speeds up to 30,000 I/sec. The average yield of the alga in this system, which uses 3 per cent cattle urine, amounts to 10 - 15 9/ m² /day. Infusion of small quantities of animal blood from a slaughter house has been found to stimulate algal growth considerably.
The alga is harvested from the sedimentation tanks and sundried.
Problems of contamination[edit | edit source]
Simple production is not the main problem in algal technology. The shallow, open-air ponds are vulnerable to contamination. Not only are methods required to maintain conditions favouring the chosen algal species to the exclusion of other organisms, but also monitoring programmes must be developed to insure a non-toxic, hygienically safe algal product. Serious problems of contamination have not been encountered, although occasional infestation of the culture with Brachionus and Chironomus larvae were observed during certain seasons. In waste stabilization ponds, contamination with other algae was frequently seen, but this could be overcome by a gradual population build-up of the desired organism.
Cultivation of algae for biofertilizer[edit | edit source]
The ability of certain forms of blue-green algae to carry out both photosynthesis and nitrogen fixation confers on them an ecological and agricultural advantage as a renewable natural resource of biological nitrogen. Nitrogen is one of the most important crop nutrients, and the great demand for nitrogenous fertilizers is apparent from the more than 580 chemical fertilizer plants now in operation or under construction throughout the world, representing an investment of over US$10,000 million.
It is estimated that the total energy required for the production of global ammonium fertilizers is equivalent to 2 million barrels of oil per day - a non-renewable resource. The energy crisis has driven fertilizer prices unrealistically high, dramatically illustrating the dependence of the world's food crops on petroleum-based fertilizers. Hardest hit by the chemical scarcity are the densely populated and land-scarce nations of Asia, where more than half of the earth's people live. Most depend on rice as their staple food. The millions of small-scale rural farmers in this region who have reaped the benefits of the new rice technology often lack the capital for chemical fertilizers. Any saving in the consumption of this fertilizer without affecting productivity, and the introduction of a cyclic nutrient supply system through biological sources, will be ecologically and economically advantageous.
Recent research has clearly shown that one of the most effective nitrogen-fixing biological systems in the rice fields are certain blue-green algae that, expressed on a per ha basis, contribute about 25 - 30 kg N/ha/season. A rural-oriented device to exploit these algae has been developed at the Indian Agricultural Research Institute at New Delhi. The merit of this process lies in its adaptability by the individual farmer without any appreciable capital investment or technical complications. Many farmers are now using this method to produce their own algal inocula for field application.
The basic principle is to grow algae using natural sunlight under conditions simulating the rice field. A thin, one-inch layer of soil is spread in rectangular trays or shallow dugout areas lined with polyethylene and flooded with two inches of water. After the soil settles down, the desired strains of blue-green algae are inoculated into these with a little superphosphate. The entire unit is kept exposed to the sun, and within a week, the entire water surface is covered by a copious growth of the inoculated species of algae. The standing water and its algae crop are allowed to dry in the sun, and the dried algal flakes are collected for field use. During bright summer, a continuous production of about 100 kg algal material is possible every fifteen days from an area of about 25 m² The cost of production is about 12 cents/kg, and the farmer needs only about 10 kg/ha to give him about 25 - 30 kg N/ha. The inherent capacity of these algae to stand extreme dessication has made it possible to preserve the product in a sun-dried form without any impairment of its viability (5, 6).
In India, a significant portion of algae production appears to have great potential as a highprotein feed supplement for livestock, particularly for poultry, and also will make an excellent biofertilizer for rice. This, coupled with the emphasis on waste recovery and efficient land utilization, will encourage the integration of algae-feed-fertilizer production with livestock raising in the nitrogen recycling systems. The major merit of algae as animal feed is that low-quality algal protein can be converted by the animals into higher quality protein in the form of meat or meat byproducts without the necessity for extensive pre-processing of the algal product. The use of algae as biofertilizer provides a cyclic nutrient-supply system with inherent ecological advantages.
References[edit | edit source]
1. H.A. Spoehr and H.A. Milner, "The Chemical Composition of Chlorella: Effect of Environmental Conditions," Plant Physiol. 24: 120 (1949).
2. All India Co-ordinated Project of Algae, Annual Report, New Delhi, 1976 - 77, p. 44; 1977 - 78, p. 72,
3. G. Clement and H. Van Landeghem, "Spirulina-ein günstiges objekt fur die Massenkultur von Mikroalgen," Ber. Deut Bot. Ges. 83 (1): 559 (1970).
4. W.E. Becker, L.V. Venkataraman and P.M. Khanum, "Digestibility Coefficient and Biological Value of the Proteins of the Alga Scenedesmus acutus Processed by Different Methods," Nutr. Rep. Internat. 14: 457 (1976).
5. G,S, Venkataraman, "Algal Biofertilizers for Rice Cultivation," Today & Tomorrow, p. 75, New Delhi, 1972.
6. G.S. Venkataraman, in W.D.P. Stewart (ed.), Nitrogen Fixation by Free-Living Microorganisms, pp. 207 - 218. Cambridge University Press, Cambridge, 1975.
Acknowledgements[edit | edit source]
Grateful acknowledgement is made to Dr. K.P. Krishnamoorthy and Mr. Jim DeVries, Projectin-Charge at Nagpur and Pondicherry centres, and to Mr. D.L.N. Rao, for their co-operation in providing necessary details, and to Dr. Ripley Fox for the culture of Spirulina platensis.
Discussion summary[edit | edit source]
Spirulina is about 90 per cent stable with sewage, and could be even more so with other substrates. There is a 30 per cent saving of inorganic fertilizer when blue green algae are used in its place to fertilize rice, and crop response is evident within one year.