This work offers a primer to the topic of AWVPs, and a description of how to build a dew collection rig. Unfortunately, dewfall is not currently recorded in most standard meteorological archives; so the next step for this project is to offer a reliable means of regional assessment that is accessible to a layman.
The problem of obtaining clean drinking water is very widespread among developing nations. The atmosphere contains an abundant amount of water in the form of vapor; however it can be difficult and costly to harvest. Atmospheric water vapor processing (AWVP) is a new field of research that is developing ways to obtain that vapor. One such method is known as radiative cooling. A surface radiates heat away until it drops below the dew point, causing moisture to condense on the surface. This is a process that occurs naturally, producing dew. The proposal is to harvest the dew that naturally forms on rooftops as a source of potable water. Rooftops made out of galvanized iron, plastic, or glass, in regions that experience the right weather conditions will naturally produce a significant amount of dew. Simply by collecting this dew, a family can gather up to 2L of dew water per dew night. This work offers a primer to the topic of AWVPs, and a description of how to build a dew collection rig. Unfortunately, dewfall is not currently recorded in most standard meteorological archives; so the next step for this project is to offer a reliable means of regional assessment that is accessible to a layman.
Introduction[edit | edit source]
As the world's population increases, fresh water supplies are being tapped out. Desalination has become a necessary means of acquiring water; however current methods are usually quite costly and use fossil fuels. This is inappropriate for the developing world, where there is a serious need for increased fresh water availability. Atmospheric water vapor processing (AWVP) is a new and emerging technology in which the atmospheric water vapor is condensed and collected.[1][2][3]
Operating Principle[edit | edit source]
There is approximately 4 g of water vapor per cubic meter of air in the earth's atmosphere. This source of potable water is available virtually worldwide. AWVPs harvest this water, by condensing it from vapor to liquid.[4][5]
Advantages[edit | edit source]
- It is at an early stage in development, but has the potential to provide environmentally acceptable alternatives to standard water supplies.[4][5]
- Many AWVP designs favor decentralization of water distribution and avoidance of huge capital costs for infrastructure.[5]
- AWVP can be made appropriate, community-managed and community-maintained for developing countries.[5]
- AWVP methods are competitive with desalination plants and simpler and less expensive to operate and maintain.[5]
- The amount of water produced would vary according to installation size, and be suitable to provide potable water to individuals or even thousands of people.[5]
- AWVP production can take place in a wide variety of locations. Thus, expensive water distribution infrastructure can be reduced or avoided.[5]
- Chemically, water vapor in the atmosphere is as clean as the air around it. Naturally occurring dew is a potable source of soft water, generally low on any mineral content.[6]
Background - Radiative Cooling[edit | edit source]

Currently, this article only considers AWVP approaches that use a cooled surface to condense water. The other common methods are using a controlled forced convection to concentrate the amount of vapor in the air, and chemical dessiants to cause it to condense.[5]
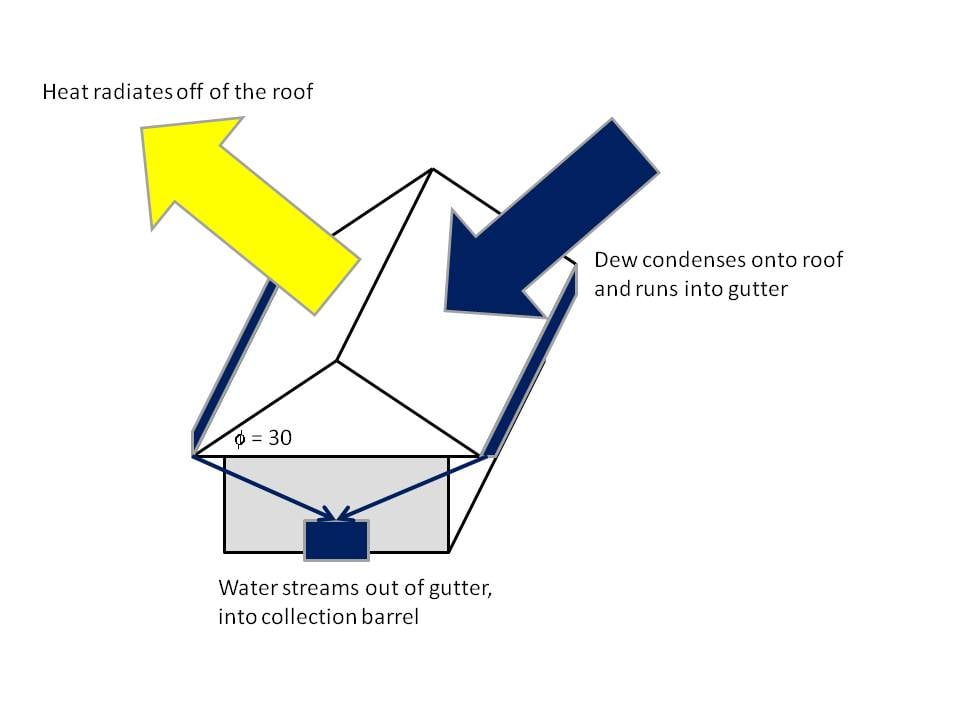
Interestingly, many desert and costal plants and animals depend on this source.[7] Naturally, the phenomenon of dew formation takes place due to night sky radiation cooling. On a clear night, heat energy is radiated away from the earth's surface into the sky. As the surface temperature drops below the dew point, water vapor will condense into droplets. Dew is formed both from water vapor in the atmosphere in the form of dewfall, as well as from water in the soil in the form of dewrise.[8][9]
The amount of dewfall can be approximated from a complex turbulent fluid analysis; however it is far simpler to consider it as an energy balance problem.[9]Note that the process of condensation releases energy.
Where
- R = net incoming radiation flux (a negative term)
- G = flux of total heat from soil to surface
- C = flux of sensible heat from air to surface
- F = flux of water vapor from air to surface
- λ = latent heat of vaporization
- Q = heat capacity of the air-grass layer per cm2
- T = mean temperature of layer
- M = heat released by vegetable metabolism per cm2
Furthermore, it follows that the maximum possible condensation rate for combined dewfall and dew-rise during nighttime is:[10][11][12][8]
Where
- Df = rate of dewfall in
- Dr = rate of dew rise in
- s = slope saturation curve in
- Q* = net radiation in
- γ = psychometric constant
- λ = latent energy for vaporization in
Studies have demonstrated that the above model can accurately and reliably predict dew rates.[8]Therefore, it is useful to quickly determine the seasonal dewfall of an area based on data from meteorological archives.
The maximum amount of dewfall possible on a given night is limited by the amount of radiative cooling possible, and is estimated to be approximately 1 mm.[11][13] In actuality, this number is usually much lower, ranging from 0.17 – 0.45 mm per night.[12]
Recent research has found materials that produce a higher dew yield. Materials that exhibit a large amount of radiative cooling will condense more dew, and hydrophilic materials will make dew collection easier. Specifically, TiO2 and BaSO4 microspheres embedded polyethylene resulted in a 20% increase of dew yield compared to Plexiglas.[14]
The angle of the surface is also important. The optimal angle has been determined through numerical models and experimental testing to be 30o from horizontal. A flatter surface will radiate more energy, however a steeper surface has improved dew collection characteristics.[14]
Unfortunately, bacterial concentrations in Dew water have been seen to exceed World Health Organization limits on various occasions. Therefore, it will require disinfectant to be considered potable.
Regional considerations[edit | edit source]
Dew collection can be useful in areas where more conventional means of water collection aren't viable, such as an arid dry region. However, dew does not form everywhere equally, and in some areas there will be nearly no dew formation at all. More work is needed to help determine a reliable and simple way for a layman to assess the seasonal dewfall in a particular area.[1]Currently, dewfall is not recorded by meteorologists, and so it can be difficult to assess the feasibility dew installations. In general, for dew to form on a given night, the following conditions must be met:[1]
- A low wind speed of approximately 2-6 m/s.
- A relative humidity of at least 30%, with higher relative humidities yielding greater amounts of dew.
- Mostly cloudless night, to allow the surface to radiate to the night sky.
Design[edit | edit source]
There are design features such as thermally insulating the roof, or building with a hyper-radiative material, would add to the dew yield. However, these things would be too costly to be economical. Instead, what is considered is an appropriate retrofit for a roof to install a dew collection system. It should be able to provide 2L per dew night from an average family sized roof.
The design is similar to that of a Rain Water Catchment System. Gutters feed water into hoses, which run to a collection barrel.
Materials[edit | edit source]
The required materials for this project are simple, and easily obtainable. Choose alternatives liberally, so long as they do not pose a risk of contaminating the water. Below is a numbered list of the required materials with bulleted alternatives.
- Roof gutter, including means of attaching it to the roof
- Standard vinyl rain collecting gutters will work and should be inexpensive and easily obtainable
- The gutter material must be hydrophilic. Something like wood would be too absorptive
- The gutter should be semi round
- Hosing
- Garden hose
- 1/4" PVC piping
- Collection Tank
- A 10L plastic food safe container
Tools[edit | edit source]
There are no specialty tools required for this project. Most of the tools needed for the construction of a solar still are common. The tools depend on what materials are being used and the level of complexity of the water collection system.
- Hacksaw
- Measuring tape
- Silicone gun
- Level
- Pliers
- Screwdrivers or hammer (for screw or nail installations)
Skills and Knowledge[edit | edit source]
For the installation, no special skills or knowledge are required. Currently however, there is no easy way for a layman to determine the usefulness of dew collation in a particular area. For this task, an engineering, or meteorology background, with knowledge of dew formation would be required.
Estimated Costs[edit | edit source]
All prices in CAD $, and are costed based on North American prices.
- Gutters cost $50 -$100 for 120 feet from sources like this
- Garden hose costs $15/50" from this source
- A container should be easily obtainable for free
In total, this project is very inexpensive. For a family sized house, with roof area of 60m2, the estimated cost would be approximately $60.
Directions[edit | edit source]
- The roofing material must be something that will not absorb any water. Ideal materials are corrugated Galvanized Iron (GI), plastic, or glass. If your roof does not have one of these materials, it will not work.
- The steepness of the roof is an important criterion. 30o from the horizontal is optimal.[14][15] The roof must have a steepness of at least 20o.
- Follow standard gutter installation practices. Traditionalroofing.com has good dirctions. The standard angle of gutter installation for rain removal is too shallow for this application. They need to be installed with a steepness of at least 2" down for every 6' across to ensure that water flows, and does not stagnate.
- The collection barrel should have a screen or net over the intake to prevent debris and insects from getting in.
Construction[edit | edit source]

- Install the rain gutters on the roof at an angle of 15o sloping away from the prevailing wind.
- Funnel low end of gutter through hosing
- Attach hosing to container
Refer to roof rain collection devices for more detail.
Case Studies[edit | edit source]
In,[1] they investigated the extent to which existing roofs given the retrofit described above can collect dew water. It was the first study of this nature done in India. The location was Kothara, India (23°14N, λ 68°45E, at 21 m a.s.l.), which is typical of rural north-western India.
The most common source of water during the dry season is wells. However, the ground water is being tapped out, receding, and becoming contaminated by salt. Dew is abundant during 8-9 months of the year. The other months are during the monsoon, where cloud cover prohibits dew formation.
Many of the local buildings use corrugated galvanized iron (GI) sheets for roofing. So, they did their testing on such a roof with an 18m2 surface area. There were 74 days during which dew formed, and a total of 113.5L of dew water collected.
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 Sharan, G., 2006, "Dew harvest: to supplement drinking water sources in arid coastal belt of Kutch," Foundation Books, .
- ↑ Rajvanshi, A. K., 1981, "Large Scale Dew Collection as a Source of Fresh Water Supply," Desalination, 36(3) pp. 299-306.
- ↑ Habeebullah, B. A., 2009, "Potential use of Evaporator Coils for Water Extraction in Hot and Humid Areas," Desalination, 237(1-3) pp. 330-345.
- ↑ 4.0 4.1 Wahlgren, R., 1993, "Atmospheric Water Vapour Processing," Waterlines, 12(2) pp. 20-22.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 Wahlgren, R. V., 2001, "Atmospheric Water Vapour Processor Designs for Potable Water Production: A Review," Water Research, 35(1) pp. 1-22.
- ↑ Beysens, D., Muselli, M., Mileta, M., 2004, "Is dew water potable? Physical, Chemical and Biological Characteristics of Dew on Atlantic Coast (Bordeaux, France), Mediterranean Coast (Zadar, Croatia) and Mediterranean Island (Ajaccio, Corsica Island, France)," Proceedings: Third International Conference on Fog, Fog Collection and Dew, Cape Town, South Africa, October, Anonymous pp. 11-15.
- ↑ Uwe G., 1979, "In the Deserts of this Earth", Harcourt Brace Jovanovich Inc.
- ↑ 8.0 8.1 8.2 Jacobs, A. F. G., Van Boxel, J. H., and Nieveen, J., 1996, "Nighttime Exchange Processes Near the Soil Surface of a Maize Canopy," Agricultural and Forest Meteorology, 82(1-4) pp. 155-169.
- ↑ 9.0 9.1 9.2 Monteith, J. L., 2007, "Dew," Quarterly Journal of the Royal Meteorological Society, 83(357) pp. 322-341.
- ↑ Monteith, J. L., 1981, "Evaporation and Surface Temperature," Quarterly Journal of the Royal Meteorological Society, 107pp. 1-27.
- ↑ 11.0 11.1 Jacobs, A. F. G., Heusinkveld, B. G., and Berkowicz, S. M., 2002, "A Simple Model for Potential Dewfall in an Arid Region," Atmospheric Research, 64(1-4) pp. 285-295.
- ↑ 12.0 12.1 Garratt, J. R., and Segal, M., 1988, "On the Contribution of Atmospheric Moisture to Dew Formation," Boundary-Layer Meteorology, 45(3) pp. 209-236.
- ↑ Jacobs, A. F. G., Van Pul, W. A. J., and Van Dijken, A., 1990, "Similarity Moisture Dew Profiles within a Corn Canopy," J.Appl.Meteorol, 29pp. 1300–1306.
- ↑ 14.0 14.1 14.2 Beysens, D., Milimouk, I., Nikolayev, V., 2003, "Using Radiative Cooling to Condense Atmospheric Vapor: A Study to Improve Water Yield," Journal of Hydrology, 276(1-4) pp. 1-11.
- ↑ Sharan, G., Beysens, D., and Milimouk-Melnytchouk, I., 2007, "A Study of Dew Water Yields on Galvanized Iron Roofs in Kothara (North-West India)," Journal of Arid Environments, 69(2) pp. 259-269.