Underbuttocks Pouch
| Health topic | Maternal mortality |
|---|---|
| Health classification | Diagnosis |
| Type | Unerbuttocks pouch |
|---|---|
| Authors | |
| Location | India |
| Status | Commercialized |
| Years |
Problem being addressed
[edit | edit source]Postpartum hemorrhaging is one of the most severe causes of maternal mortality. PPH (Postpartum hemorrhaging) rates in the developed worlds have severely decreased, but they are still a great threat in developing countries. The World Health Organization suggests that there are over 100,000 deaths due to postpartum hemorrhaging every year.
Detailed description of the solution
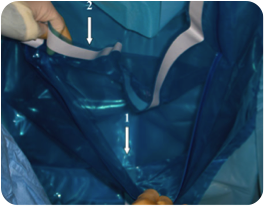
[edit | edit source]This device is classified as one which helps in PPH estimation. This device consists of a single use conical calibrated pouch incorporated into an underbuttocks drape type 3M™ Steri-Drape™ 1084. The pouch is placed in sterile conditions at the start of the expulsion phase. The first compartment (of two) is used at during delivery to collect the amniotic fluid and the second is positioned immediately after the birth to evaluate blood loss.
Designed by
[edit | edit source]- Designed by: Microtek
When and where it was tested/implemented
[edit | edit source]Funding Source
[edit | edit source]References
[edit | edit source]Other internally generated reports
[edit | edit source]Externally generated reports
[edit | edit source]Tixier, H., et al., Interest of using an underbuttocks drape with collection pouch for early diagnosis of postpartum hemorrhage. Archives of Gynecology and Obstetrics, 2011. 283(1): p. 25-29.
IP and copyright
[edit | edit source]Approval by regulatory bodies or standards boards
[edit | edit source]| Authors | |
|---|---|
| License | CC-BY-SA-3.0 |
| Cite as | Atessa (2012–2024). "Underbuttocks Pouch". Appropedia. Retrieved June 4, 2026. |