Self-Administered Adhesive
| Map | |
|---|---|
| Location | Israel |
| Coordinates | 30° 48' 44.73" N, 34° 51' 34.11" E |
| Health topic | Tuberculosis |
|---|---|
| Health classification | Diagnosis |
Problem being addressed
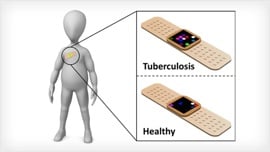
[edit | edit source]Tuberculosis (TB) infects more than 8 million people per year, making its swift diagnosis and treatment critical to prevent the spreading of the disease. Current diagnosis methods are often inaccurate and are time intensive, taking up to 8 weeks for a result. The inaccuracy and long diagnosis times of these methods create a risk for an infected person to spread the disease before receiving the proper treatment.
Detailed description of the solution
[edit | edit source]The self-administered adhesive plaster method relies on volatile organic compounds (VOCs), which are emitted from infected cells and their surrounding microenvironment. These VOCs can be detected due to the changes they cause in the skin and through their exchange via the blood. Organic sensors that detect these changes placed in stable adhesive plasters can be put directly on the chest, making the diagnosis process less time consuming and potentially more accurate.
Designed by
[edit | edit source]- Designed by: Technion, Israel Institute of Technology (primary investigator Hossam Haick as of May 2012).
- Manufacturer (if different): n/a
- Manufacturer location: n/a
Funding Source
[edit | edit source]Received grant from Grand Challenges in Global Health as of May 2012.
References
[edit | edit source]Other internally generated reports
[edit | edit source]- "Self-administered adhesive plaster for detection of tuberculosis" (Jan 14, 2014). Retrieved on Jul 16, 2014 from http://webcache.googleusercontent.com/search?q=cache:Nr9qXCn5Qm4J:map.technion.ac.il/.upload/Rotem%2520Ermanok%252014%2520Jan%25202014_yt.doc+&cd=1&hl=en&ct=clnk&gl=us
Externally generated reports
[edit | edit source]- "Self-administered adhesive plaster for detection of tuberculosis" (2012). Retrieved on Aug 6, 2014 from http://www.grandchallenges.org/SiteCollectionDocuments/map/iframes/grants-print.aspx?topics=Agriculture;Cell%20phone%20applications;Aid%20is%20Working;Contraception;Data%20systems;Diagnostics;Drug%20resistance;Global%20health%20solutions;HIV%20infection;Malaria%20eradication;Anti-malarial%20Compounds;Mothers%20and%20newborns;Mucosal%20immunity;Neglected%20tropical%20diseases%20;Nutrition;One%20Health;Poliovirus%20eradication;Protective%20strategies;Sanitation;Synthetic%20biology;Tuberculosis%20latency;Vaccines;&challenge=&date=2012;&continent=&phase=1;.
Photo Source
[edit | edit source]Retrieved on Jul 30, 2014 from http://www.grandchallenges.org/SiteCollectionDocuments/Photo%20Gallery/photogallery_v2.aspx?group=Grand%20Challenges%20Explorations%20Round%208%20Grants#go.
| Authors | |
|---|---|
| License | CC-BY-SA-3.0 |
| Cite as | Dhodg (2014–2023). "Self-Administered Adhesive". Appropedia. Retrieved June 4, 2026. |