
Rice hulls are unique within nature. They contain approximately 20% opaline silica in combination with a large amount of the phenyl propanoid structural polymer called lignin. This abundant agricultural waste has all of the properties one could ever expect of some of the best insulating materials. Recent ASTM testing conducted R&D Services of Cookville, Tennessee, reveals that rice hulls do not flame or smolder very easily, they are highly resistant to moisture penetration and fungal decomposition, they do not transfer heat very well, they do not smell or emit gases, and they are not corrosive with respect to aluminum, copper or steel. In their raw and unprocessed state, rice hulls constitute a Class A or Class I insulation material, and therefore, they can be used very economically to insulate the wall, floor and roof cavities of a super-insulated Rice Hull House. This paper also explains how the structure of such a house can be fashioned out of a variety of engineered lumber products derived from sugarcane rind.
Paper[edit | edit source]
When nature decided how to package a grain of rice, she wrapped this tiny bundle of nutrients with what is often referred to as a "biogenic opal."[1] The chemical structure of the rice hull, containing amorphous silica bound to water, closely resembles that of the opal, and this gives the rice hull some fairly amazing properties. Nowhere could we ever find a cereal by-product so low in protein and available carbohydrates, and yet, at the same time, so high in crude fiber, crude ash and silica.[2] Of all cereal byproducts, the rice hull has the lowest percentage of total digestible nutrients (less than10%).[3]

The rice hull contains approximately 20% opaline silica in combination with a large amount of the phenyl propanoid structural polymer called lignin. Such a high percentage of silica is very unusual within nature,[4] and this intimate blend of silica and lignin makes the rice hull not only resistant to water penetration and fungal decomposition, but also resistant to the best efforts of man to dispose of it. Since rice is grown on every continent except Antarctica, since it ranks second only to wheat in terms of worldwide area and production,[5] and since the hull represents on average about 20% of the rough harvested weight of rice,[6] our planet ends up with an abundance of this scaly residue.
More than 100,000,000 metric tons of rice hulls are generated each year throughout the world.[7] In 1995, the United States produced about 1,260,000 metric tons of rice hulls[8] at about 50 mills[9] located in Louisiana, Texas, Arkansas, Missouri, Mississippi, Florida and California. Since most mills store rough rice and process it on a daily basis, fresh dry hulls are available throughout the year. Since hulls do not biodegrade or burn very easily, they are sometimes available free-of-charge.
- Husks typically sell for about $6/ton, although one mill indicated that it has sold husks for prices ranging from $2 to $20 per ton.[10]
The hull is a very tough and abrasive packaging material, consisting of two interlocking halves. It encapsulates the tiny space vacated by the milled grain, and in proximity to a myriad of other hulls, it forms a thermal barrier that compares well with that of excellent insulating materials.[11] Thermal resistance tests on whole rice hulls indicate R-values greater than 3.0 per inch.[12] If the R-value of rice hulls is so favorable, why have they not been used extensively to insulate residential and commercial structures?[13]

Perhaps our scientists and engineers focus only on creating materials and products that can be labeled and marketed as proprietary. Perhaps the humble use of the rice hull as an insulation material does not sufficiently inspire the scientific or commercial imagination. But why focus on man-made products when natural materials abound? Surely there must be some profound and obvious reason that makes the raw rice hull unsuitable to serve as an insulation material.
Do rice hulls burn? Yes they do, but with difficulty, as Eldon Beagle once so elegantly explained:
- "The peculiar silica-cellulose 'drinking-straw bundle' structural arrangement of the husks results in an object that does not burn or even liberate heat in a manner resembling that of any organic substance. These minute silica-crested tubular structures offer an inherent resistance to burning. Often they seal off and prevent the thorough, uniform burning essential to obtaining a desired end-product."[14]
Anyone who has tried to set a match to loose rice hulls understands how difficult they are to burn. Since air cannot flow freely through a pile of rice hulls to provide the oxygen needed to sustain rapid combustion, they do not easily and cleanly combust. The bulk density of loose rice hulls is similar to that of baled straw, and anyone who has tried to burn a bale of straw understands the problem associated with the availability of oxygen. But the simple availability of oxygen does not explain everything.
As we have noted above, the high percentage of opaline silica within rice hulls is most unusual in comparison to other plant materials, and some scientists say that during the combustion of rice hulls, the silica ash may form a "cocoon" that prevents oxygen from reaching the carbon inside. Other scientists speculate that, since silica and carbon may be partially bonded at the molecular level, silicon carbide is formed during high-temperature combustion, and that the presence of this heat-resisting ceramic impedes the easy combustion of the rice hull.[15] Still other scientists say that at certain temperatures, the molecular bond between the silica and carbon in the hull is actually strengthened, thereby preventing the thorough and uniform burning of the hull.[16] In any case, even if we do manage to ignite a pile a rice hull, we find that it tends to smolder rather than flame.
- Rice husks are flame retarding and, at ordinary temperatures, selfextinguishing. A lighted match, tossed onto a pile of rice husks will generally burn out without producing a self-sustaining flame in the husks.[17]
Conventional cellulose insulation necessitates the addition of large quantities of flame and smolder retardants. The concentration of flame and smolder retardant chemicals (such as boric acid, sodium borate, ammonium sulfate, aluminum sulfate, aluminum trihydrate, mono- or di-ammonium phosphate) in conventional cellulose insulation may reach as high as 40% by weight.[18] These chemicals are expensive to purchase and prepare, and the cellulosic fiber must undergo extensive preparation to receive them.
Surprisingly, rice hulls require no flame or smolder retardants. Nature has freely given to this agricultural waste product all of the combustion properties needed to pass the Critical Radiant Flux Test (ASTM C739/E970-89), the Smoldering Combustion Test (ASTM C739, Section 14), and the Surface Burning Characteristics Test (ASTM E84). Recent testing done by R&D Services indicates an average Critical Radiant Flux (CRF) of 0.29 W/cm2, a smoldering combustion weight loss between 0.03% and 0.07%, a Flame Spread Index (FSI) of 10 and a Smoke Development Index (SDI) of 50. Since US building codes require an FSI of 25 or less, and an SDI of 450 or less, we see that the rice hull easily passed these tests. In its raw and unprocessed state, the rice hull constitutes a Class A or Class I insulation material.
All organic materials will absorb or release moisture until they come into equilibrium with the relative humidity of the surrounding air. The high concentration of opaline silica on the outer surface of the rice hull impedes the atmospheric transfer of moisture into the hull. Also, 2.1% to 6.0% of the rice hull consists of a biopolyester called cutin,[19] which, in combination with a wax produced by the rice plant, forms a highly impermeable barrier. Nature employs several very effective strategies to protect the kernel of rice from the water and high humidity generally associated with the cultivation and growth of this plant.
Consequently, studies done on rice hulls at 25°C indicate that the equilibrium moisture content of rice hulls at 50% relative humidity is at or below 10%, while at 90% relative humidity, the equilibrium moisture content of rice hulls remains at or below 15%.[20] A Moisture Vapor Sorption Test (ASTM C739, Section 12) conducted by R&D Services indicates a gain in weight of only 3.23%. This is well below the moisture content needed to sustain the growth of fungi and mold.
The ASTM Standard Specification for cellulose insulation requires a 28-day test for resistance to fungal growth (see section 10 of ASTM C1497, ASTM C1338, Section 6.6 of ASTM C1149 or Section 11 of ASTM C739). Following these standards, R&D Services inoculated rice hulls with five different fungal species, and the rice hulls passed these tests without the addition of fungicides or any other chemicals.
The high concentration of opaline silica on the outer surface of the rice hull also establishes the effective hardness of the rice hull at roughly the same values as reported for opal (6 on the Mohs scale).[21] However, due to the presence of lignin within the rice hull, this hardness is tempered with flexibility and elasticity. Since the rice hull is hard and yet elastic, it resists settling and compression far better than shredded newspapers. The settling of cellulose insulation in a wall cavity can reduce its installed height by as much as 25%. For this reason it is often necessary to stabilize cellulose insulation by means of polyvinyl acetate or an acrylic adhesive. None of these stabilizing compounds are needed with rice hulls, if firmly vibrated or packed into a wall cavity.
Ordinarily loose rice hulls have an angle of repose of about 35 degrees.[22] But once firmly packed into a wall cavity, their tiny tips, edges and hairs interlock to achieve a negative angle of repose. Due to this peculiar bonding of rice hulls under mild pressure, they stabilize in a very uniform manner, and no further settling is possible. Also, since it is not necessary to add fire-retardants, fungicides or any other chemicals to the rice hull, R&D Services has determined that this benign and stable biomass does not emit offensive odors (ASTM C739). Likewise, R&D Services determined that rice hulls do not corrode aluminum, copper or steel (ASTM C739, Section 9).
With rice hulls, we need not engage in a mining or manufacturing process that generates air pollution, water pollution or erosion.[23] With rice hulls, we need not engage in a manufacturing process that depletes our reserves of fossil fuels (as with polystyrene,[24] polyisocyanurate and polyurethane insulation). With rice hulls, we do not use chlorinebased chemicals such as phosgene, propylene chlorohydrin[25] or any ozone-depleting chlorofluorocarbons.[26] With rice hulls, we do not use urea formaldehyde, and surely none of the phenol formaldehyde used in most fiberglass insulation.[27] With rice hulls, we do not have to worry about the irritability or cacinogenicity of dust and fibers.[28] Moreover, those with acute chemical sensitivity should not have to worry about the offgassing associated with binders in batt insulation, with ink in recycled newspaper or with VOCs released from foam insulation.[29] Since rice hulls require no shredding, hammer-milling, fluffing, fiberizing, binding or stabilizing, they possess, surely in those states where hulls are available, far less embodied energy than even cellulose insulation.[30] Since rice hulls do not burn very easily, they require no flame or smolder retardants, and since they are so tough and durable, nothing prevents them from being used and recycled over and over again.
Perhaps the most significant cost associated with the utilization of the rice hull is its transport. At a bulk density of about 9 lbs. per ft3,[31] loose hulls can be transported at roughly the same cost as baled straw. However, to reduce the cost of transport, rice hulls can be compressed to as much as 25 lbs. per ft3 without destroying their elasticity.[32] They readily bounce back to their original density once the force of compression is removed. But to transport rice hulls economically, it would not be necessary to compress rice hulls to a density of 25 lbs/ft3. At a density of only 14.50 lbs/ft3, a standard 53-foot trailer attains optimal transport efficiency at its maximum legal weight of 24 tons. If, at this transport density, we pay an average trucking fee of $1.45 per mile, it would cost approximately $15, $30, $45, $60, $75 and $90 to transport one ton of rice hulls 250, 500, 750, 1000, 1250 and 1500 miles respectively (see graph below).
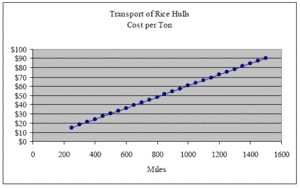
At an installed density of 9 lbs/ft3, one ton of rice hulls will insulate 222 ft2 of a 12-inch wall cavity. Therefore, the cost per ft2 incurred by transport over these same distances is $0.07, $0.14, $0.20, $0.27, $0.34 and $0.41 respectively (see next graph).
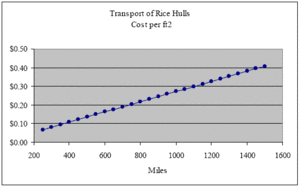
Those living less than 200 miles from rice mills should have a hard time justifying the use of any other type of insulation material. When many mills reluctantly sell rice hulls for less than $5.00 dollars per ton, the argument in favor of rice hulls becomes even more compelling. At $5.00 per ton, the cost of rice hulls per ft2 of a 12-inch deep wall is a mere $0.02.
Supposing that we pay not $5.00 but $25 per ton (well above current market value), we find that the purchase price of the rice hulls per ft2 wall insulated is only $0.11. Adding this $0.11 back into the cost of transport over these same distances, we arrive at a total cost per ft2 of the rice hull delivered to the job site of $0.18, $0.25, $0.32, $0.38, $0.45 and $0.52 respectively (see graph below).
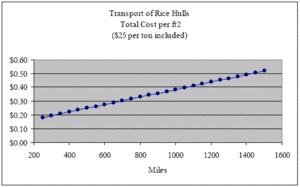
With these simple calculations, we see that the transport of rice hulls should not limit or constrain their widespread use as insulation. These calculations allow us to make two comparisons, one with respect to baled straw and the other with respect to dense-pack cellulose insulation. Relative to all other types of insulation on the market today, these two types of insulation possess the highest recycle content and the lowest content of embodied energy.
On average, a two-string straw bale (14x18x36 inches) weighs 45 pounds, sells for $2.50, and transports to the job site for an additional $1.00.[33] Laid flat within a wall, the twostring bale represents 3.5 ft2 of wall surface. This gives a purchase price of $0.71 per ft2 of wall, to which we must add another $0.29 for transport. Accordingly, the total cost of baled straw per ft2 of bale wall is approximately $1.00. This represents more than five times the price of the rice hull transported 250 miles and almost twice the price of the rice hull transported 1,500 miles. Moreover, 12 inches of rice hulls at R-3.0 per inch deliver 37% more insulation than 18 inches of baled straw at R-1.45 per inch,[34] and this, at one fifth to one half the cost, using 33% less wall space.
Cellulose insulation in a dense-pack application is inserted into a wall at a density of approximately 3.5 lbs/ft3. Accordingly, one ton of cellulose insulation will insulate 571 ft2 of our proposed 12-inch deep wall. At an average delivered price of $540 per ton, cellulose insulation costs roughly $0.95 per ft2 of wall insulated. This is slightly cheaper than baled straw, but still roughly five times the price of the rice hull transported 250 miles and twice the price of the rice hull transported 1,500 miles.
If rice hull insulation compares well with straw bale and cellulose insulation, then how much more desirable should it be than those forms of insulation of a low recycle content and a high embodied energy content? The building industry in the United States demands several million tons of insulation on an annual basis. Should rice mills not form an alliance with architects and builders in displacing all forms of insulation not produced in an environmentally efficient and beneficial manner?
- In rebuttal, someone might argue quite correctly that a load-bearing straw bale wall provides a lot more than just insulation. Someone might also argue that we have compared the theoretical insulation value of rice hulls over and against the installed insulation value of straw bales – a classic case of apples and oranges. But insofar as the wall system is correctly designed (no thermal conductivity via structural members), and insofar as the rice hulls are uniformly distributed and packed within the wall (no spaces unoccupied with hulls), the theoretical and installed values should be the same.
In 1994, the cellulose insulation industry consumed 420,000 tons of recycled newspapers.[35] This is an activity that should be promoted and enhanced in every possible way. If rice mills were to contribute an equivalent amount to the insulation industry, this would constitute only one third of the annual production of rice hulls in the United States.
At an average selling price of $25 per ton and an average transport distance of 600 miles, this would generate each year roughly $10.5 million dollars of revenue for rice mills and over $15 million dollars for transport companies. Since the United States generates less than 1.3% of the global production of rice hulls, the rest of the world has a lot more to gain from this simple and unsophisticated use of such an abundant agricultural byproduct. Supposing we are convinced that rice hulls offer many advantages over conventional insulating materials, how should we go about constructing a super-insulated house employing rice hulls? Supposing further that we wanted to build the structure of this house almost entirely out of agricultural waste materials, how should we proceed? Since loose rice hulls, unlike bales of straw, have no structural value, how should we build the floor, wall and roof cavities of a rice hull house? The technology that would allow us to create floor, wall and roof systems out of lowgrade cellulosic materials already exists. Companies such as Georgia-Pacific, Louisiana Pacific, Weyerhaeuser and Boise specialize in a variety of engineered lumber products such as I-joists, laminated veneer lumber (LVL), parallel strand lumber (PSL), laminated strand lumber (LSL), oriented strand lumber (OSL), glued laminated timber (GLULAM), etc. These engineered wood products offer a broad range of advantages over traditional solid-sawn lumber.
They are free of knots and other imperfections. They do not shrink, crown, twist, bow, split, check or warp. They are stronger, stiffer, lighter, straighter and far more precise than solid-sawn lumber. They can be engineered to span relatively long distances, with far more load-carrying capacity per unit of weight. Architects can design structures with far more livable and useful space, builders are not confronted with discard and waste, and carpenters find them easy to cut and install.
Currently, aspen is the primary raw material used to manufacture many of these engineered lumber products. Aspen grows in forests, and up until now, the destruction of these forests has been an inevitable consequence of making engineered lumber. Strawbale construction grabs our attention in such a powerful way for the simple reason that it makes use of an abundant agricultural waste material. In constructing a load-bearing strawbale wall, we need not chop down a single tree or impact in any way the abundant and diverse life that depends on trees. Moreover, we need not tie up an additional acre of farmland in producing the bales that are needed for its construction. The bale of straw is a secondary product or by-product of growing wheat and making flour.
Just as agriculture gives us the rice hull as a by-product of rice, can agriculture not also give us a strong woody fiber as a by-product of something else? We are looking for an agricultural by-product, that, as if by some internal logic of nature, should resemble bamboo in many ways. Not only should it be fast-growing and strong, but we might even expect to find a remarkable similarity to bamboo in terms of the structure of the plant itself, more specifically, the series of nodes and internodes that characterize the stalk or culm. Like bamboo, it should be a tall perennial grass possessing a thick-walled outer rind or cortex, but unlike bamboo, we might hope that it could be harvested and utilized as a mere by-product of something else. No land would have to be set aside specifically for its cultivation, and all of the equipment needed for its harvesting, transportation and pre-processing should already be in place. Where should we go to find such an amazing plant?
It already exists. Introduced by Christopher Columbus to the New World as far back as 1493, sugarcane represents nothing new to agriculture within the United States.[36] But ever since its introduction to the Americas, it has been cultivated primarily for the large amount of sucrose that fills its inner core. Even though all high-value sucrose lies primarily in its interior, the entire stalk is crushed, pressed and processed, and in so doing, the integrity, strength and value of the woody outer rind is totally destroyed. At best, it serves as a low-grade fuel, and at worst, it is discarded outdoors in large piles where it might eventually self-ignite through the action of thermophilic bacteria. It is hard to understand just what we are throwing away when we incinerate the outer rind of the sugarcane plant. If we compare the growth rate and fiber quality of a typical forest in the Pacific Northwest to that of a typical sugarcane plantation in Louisiana, we would discover to our great amazement that the sugarcane field clearly outperforms the forest by a large margin. Within an equivalent period of time, one acre of sugarcane can produce almost twice the board feet as an acre of the forest.
Once the whole stalk or billet passes through a conventional mill press, not only is the structural integrity of the rind destroyed, but the pith and rind become intimately mixed, and any possibility of economically separating them is destroyed. The inner core or pith of the sugarcane plant represents a biodegradable hemicellulosic fiber that has very little structural value, and if we are looking for alternatives to conventional forest products, even a small amount of pith in manufactured wood products would be quite undesirable. All attention, therefore, shifts to finding the means to separate rind from pith prior to the cutting and grinding of the sugarcane at the mill.
Today most cane in developed countries is harvested by means of a billet harvester. The billet harvester knocks the stalk down and bites into the surface of the row, taking along a lot of trash such as tramp iron, sand, clay, clay balls, stones, bricks, leaves and tops. The harvester then cuts the stalk into billets approximately 8 inches in length. In the best case, under dry conditions, a ton of cane contains 8% inorganic trash by weight, and under wet conditions, a ton of cane contains up to 30% inorganic trash by weight. Not only is the tough cellulosic fiber of the cane totally destroyed in the grinding process at the mill, but this fiber is intimately mixed at times with equal amounts of inorganic debris.
This trash going into the mill contains no sugar, and as it leaves the mill in the form of filter cake or bagasse, it carries away sugar. Each one percent of trash in the billets represents a loss in production of three pounds of sugar per ton of cane. More trash means more maintenance, more flocculent, more lime, more natural gas, more unburned bagasse, more filter cake, more front-end loaders, more water treatment, more settling basins, more draglines to clean out settling basins, more haulage, more inversion, more molasses, less sugar and more cost. Surely there has to be a way to maximize the recovery of sugar and minimize the cost of producing it, while, at the same time, fully preserving the integrity of the rind.
To solve this problem, two types of separation are required. The first separator, a dense medium separator, removes all inorganic debris from the billeted cane, and a second separator, a mechanical separator, separates pith from rind.
Engineering, Separation and Recycling LLC of Washington, Louisiana, has designed over 20 dense medium separators used extensively in the pre-processing of a large variety of root vegetables, such potatoes, carrots, salsifies and beets. This same vegetable separator can be used very effectively to separate billets from inorganic debris (see picture below). Since a sugar cane billet has a density of about 1.09 RD (quite similar to that of a potato), and since clay balls, stones, and bricks have a density well above 2.00 RD, this separation is easy and straight-forward.

After all the inorganic material is removed from the billets, they are then routed to a very modern and sophisticated mechanical separator called the "Tilby Separator." The Tilby Separator consists of an initial set of rollers (a splitter station) where the billets are cut into two longitudinal halves. Afterwards, the process splits into a right and a left set of rollers (a depithing station), each set scraping out and separating the pith from the rind. The billets pass through the splitter and depithing stations at an extraordinary speed of 20 feet per second. Only the pith is routed to the juice extractor, while the rind is sent to a dryer. Once dried to a moisture content less than 2%, the rind can be fabricated into just about any type of engineered lumber imaginable.
Since the fiber structure and length of the original billet are fully preserved in the Tilby process, a lot less glue is required in the manufacture of engineered lumber than would ordinarily be the case. If fashioned into engineered wooden studs, we have all that would be needed to create the wall cavities of our proposed rice hull house. If fashioned into engineered-wood-product I-joists, we have all that we need to create floor and roof cavities. Sills, floor tiles, roof shakes, even the fiber needed in the manufacture fibercement siding, could all be made from the extremely robust fiber of the sugarcane rind.[37][38]

Through the use of engineered lumber, the option of raising the entire structure off the ground (pier-and-beam) presents itself at a reasonable cost and permits siting in lowlying areas or on uneven terrain. Similarly, the attic easily converts into open, unobstructed and useful living space, greatly reducing the average cost per square foot of the structure.[39] Since the world-wide annual production of raw sugarcane situates at about one billion tons, about 75 million tons of dry rind are potentially available to the engineered lumber industry. This just about matches the 100 millions tons of rice hulls available each year throughout the world. With these two by-products of sugar and rice, we can build and insulate millions of homes each year.
As citizens in a large industrial society, we find it hard to do things that really make a difference. In choosing to build a structure derived in large measure from agricultural waste materials, not only do we do that which is right with respect to the environment, but we also fashion for ourselves a structure far superior to anything conventionally available. This super-insulated rice hull/sugarcane house, when correctly designed,[40] should be considerably cheaper to build than a conventional structure, while continuously benefiting its owner with utility bills never exceeding on average more than one US dollar per day.
Therefore, why build conventionally when it is far cheaper and far more sensible in every respect to do otherwise? Up until now, we could always take refuge in the fact that as long as we are unaware of the possible, we could not make it happen. But this is not so much about duty and obligation, as it is about finding new and exciting ways of responding creatively to the awesome and overwhelming beauty within our Universe.
Appendix[edit | edit source]


The first rice hull house, completed February, 2004, is the home of Paul and Ly Olivier. Located in the historic steamboat town of Washington, Louisiana, right across from the magnificent Magnolia Ridge Plantation,[41] it is indistinguishable from houses built in the area more than 150 years ago. Many of the building techniques described in this paper have been applied in the construction of this home.
Paul Olivier
Engineering, Separation & Recycling LLC
P.O. Box 250
Washington, Louisiana 70589
Telephone: 1-337-826-5540
Email: xpolivier@hotmail.com
Notes[edit | edit source]
- ↑ Velupillai, L., Mahin, D.B., Warshaw, J.W., and Wailes, E.J. 1996. A Study of the Market for Rice Husk-to-Energy Systems and Equipment, p.24, Louisiana State Agricultural Center. "In nature, silica (SiO2) occurs as seven distinct polymorphs: quartz, cristobalite, tridymite, coesite, stishovite, lechatelerite(silica glass), and opal; the latter two are amorphous." Drees, L., Wilding, L., Smeck, N., and Senkayi, A.1989. Minerals in Soil Environments (2nd Edition), p.913, "Opal is a hydrated silica polymorph(SiO2.nH2O)." Ibid, p.921
- ↑ Rice-Husk Ash Cements: Their Development and Applications, United Nations Industrial Development Organization, Vienna, pp.12-13
- ↑ Juliano, B.1985. Rice: Chemistry and Technology, p. 695
- ↑ "No other plant offal even approaches the amount of silica found in rice husks." Beagle, E.C. 1978. FOA Agricultural Services Bulletin 31, p.8
- ↑ Velupillai (1996), p.1
- ↑ ibid., p.15. See Beagle (1978), pp.6. "Percentages of husk in paddy vary widely, but 20% can be taken as a fair average." Ibid, p.25
- ↑ Velupillai (1996), p.15
- ↑ ibid., p.44
- ↑ ibid., p.37. For a listing of some rice mills in the United States, see http://web.archive.org/web/20060312140454/http://www.ricecafe.com:80/newlinks2.htm (deleted site; nov 2010) or ftp://www.usarice.com/publish/member1.htm (requires membership; nov 2010)
- ↑ Velupillai (1996), p.45
- ↑ Velupillai (1996), p.16
- ↑ "Rice hull has a thermal conductivity of about 0.0359 W/(m.°C); the values compare well with the thermal conductivity of excellent insulating materials (Houston, 1972)." Juliano (1985), p.696. The thermal conductivity of rice hull ash is reported to be 0.062 W.m-1.K-1. See UNIDO, p.21. A more recent test done by R&D services of Cookville, Tennessee, indicates a 3.024 R-per-inch.
- ↑ Although charred rice hulls have been sold as an insulation material in loose-fill applications under the trademark name of "Mehabit," it is hard to find evidence that fresh hulls have been used for this purpose. See Beagle (1978), p.132
- ↑ Beagle (1978), p.8. "The high percentage of silica in rice hulls and the peculiar silica-cellulose structure impede uniform and thorough burning of the hulls in a combustion process." Velupillai (1996), p.18. "Of all biomass combustion, the combustion of rice hulls (and straw) is particularly difficult because of the high ash content." Ibid., p.23. "Eldon Beagle set a pile of rice hulls 300'x500'x50' on fire and they burned forsix months." Ibid., p.24. "However, husk cannot be burnt easily or cleanly with excess air, and energyrecovery is very low as the heat produced cannot be utilized in a beneficial manner." Ibid., p.25
- ↑ ibid., p.24
- ↑ From a conversation with Carl D. Simpson of Riceland Foods, Inc
- ↑ Beagle (1978), p.9, quoted from Burrows (109A)
- ↑ "The concentrations of chemicals commonly added in commercial cellulosic insulation normally range from 10 to 40% by weight. Chemicals commonly used are boric acid, sodium borate, ammonium sulfate, aluminum sulfate, aluminum trihydrate, mono- or di-ammonium phosphate." Service Bulletin entitled"Borates for Fire Retardancy in Cellulosic Materials," p.5, prepared by US Borax
- ↑ Juliano (1985), p.695. Regarding cutin[1] (no such page, nov 2010)
- ↑ Juliano (1985), p.707
- ↑ Juliano (1985), p.696
- ↑ Juliano (1985), p.28
- ↑ Much of the comparative language of this paragraph is taken from Environmental Building News – Insulation Materials: Environmental Comparisons[2]
- ↑ "The styrene used in polystyrene insulation is identified by the EPA as a possible carcinogen, mutagen, chronic toxin, and environmental toxin. Further, it is produced from benzene, another chemical with both environmental and health concerns." Ibid, p.5
- ↑ "To manufacture isocyanate, a precursor of polyisocyanurate and polyurethane insulation, two chlorine based chemicals are used: phosgene and propylene chlorohydrin." Ibid., pp.4-5
- ↑ "The most significant pollutants found in insulation materials are chlorine-based chemicals that destroy the earth's protective ozone layer." Ibid., p.5
- ↑ "Most fiberglass insulation is produced using a phenol formaldehyde (PF) binder to hold the fibers together." Ibid., p.5
- ↑ "Growing health concerns about glass fiber" are discussed on p.10 of ibid
- ↑ ibid., pp.10-11
- ↑ Embodied energy is defined as "the energy required to produce and transport materials." Ibid., p.8
- ↑ Juliano (1985), p.696, Velupillai (1996), p.16, Beagle (1978), p.8
- ↑ "Hulls can be readily compressed to about 0.4 g/cm3, and grinding increases bulk density two to four times." Juliano (1985), p.696
- ↑ These figures were submitted by Catherine Wanek, editor of the popular straw bale publication called The Last Straw
- ↑ "Measurements then showed the (straw bale) wall to insulate to R-27.5 (RSI-4.8). On a per-thickness basis, this is R-1.45 per inch (0.099 W/m°C), just about over half of the value most commonly reported."[3] p.2
- ↑ www.buldinggreen.com p.2 (subscription needed, nov 2010)
- ↑ http://web.archive.org/web/20020316053751/http://www.lib.lsu.edu:80/special/exhibits/sugar/case1.html (link not working, nov 2010)
- ↑ For more information on this rice hull construction technique[4]
- ↑ The Elimination of Extraneous Material from Sugarcane Billets [5]
- ↑ If, for example, the downstairs represents 60% of the total living space at a cost of $80/ft2, and if the upstairs can be converted into 40% of the total living space at an additional cost of $10/ft2, then the average cost/ft2 is only $52
- ↑ "Thick enough insulation and good enough windows can eliminate the need for a furnace, which represents an investment of more capital than those efficiency measures cost. Better appliances help eliminate the cooling system, too, saving more capital cost. The only moderately more efficient house and car do cost more to build, but when designed as whole systems, the superefficient house and car can often cost less than the original, unimproved versions." Hawkens, P., Lovins, A., and Lovins, H. 1999. Natural Capitalism, p. 114, Boston: Little, Brown and Company
- ↑ http://web.archive.org/web/20050624085458/http://www.cajuntravel.com:80/washington.html