Problem being addressed[edit | edit source]
8% of all maternal deaths in developing countries are due to obstructed and prolonged labor. Obstructed labor can occur because of cephalopelvic disproportion, abnormal presentations of fetal anatomy and certain abnormalities of the reproductive tract.
Detailed description of the solution[edit | edit source]
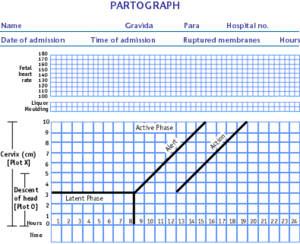
The partograph improves obstructed labor outcomes by providing obstetricians or midwives with a pictorial overview of the labor's progression, which makes early identification of an obstructed labor possible and gives them more time to manage the obstructed labor. The partograph may include measurements such as cervical dilation over time, fetal heart rate and vital signs and helps health care professionals to detect deviations from normal labor. The only costs associated with the partograph are printing and training costs.
Designed by[edit | edit source]
- Designed by: World Health Organization
- Manufacturer (if different):
- Manufacturer location:Geneva, Switzerland.
When and where it was tested/implemented[edit | edit source]
There are many versions of the partograph available today. Friedman's labor curve is a crude version that is used by many midwives and healthcare professionals. Friedman's labor curve is sensitive to interference, prolonged labors with sedation and shortened labors. Philpott's partograph is an improved version of Friedman's labor curve and was developed to meet maternal health needs in Harare, Zimbabwe. Philpott's partograph has a curved alert line that allows prolonged labor to be managed more effectively. The WHO developed its own partograph as an adaptation of the Philpott partograph. The WHO developed and evaluated its version of the partograph in 1994. A second version of it was created in 2000, removing the latent phase.
Funding Source[edit | edit source]
World Health Organization
References[edit | edit source]
Peer-reviewed publication[edit | edit source]
- Mathews, J. E., Rajaratnam, A., George, A., & Mathai, M. (2007). Comparison of two World Health Organization partographs. International Journal of Gynecology & Obstetrics, 96(2), 147-150.
- Mathai, M. (2009). The partograph for the prevention of obstructed labor. Clinical Obstetrics and Gynecology, 52(2), 256-269.
- Chongsuvivatwong, V., & Fahdhy, M. (2005). Evaluation of World Health Organization partograph implementation by midwives for maternity home birth in Medan, Indonesia. Midwifery, 21(4), 301-310.
- Petterson, K. O., Svensson, M. L.,& Christensson, K. (2000). Evaluation of an adapted model of the World Health Organization partograph used by Angolan midwives in a peripheral delivery unit. Midwifery, 16(2), 82-88.
- Lennox, C. E., Kwast, B. E., & Farley, T. M. (1998). Breech labor on the WHO partograph. International Journal of Gynecology & Obstetrics, 62(2), 117-127.
- (1994). World Health Organization partograph in management of labour. Lancet, 343(8910) 1399-1404.
Other internally generated reports[edit | edit source]
- Soni, B. L. (2009). Effect of partogram use on outcomes for women in spontaneous labour at term: RHL commentary. The WHO Reproductive Health Library.