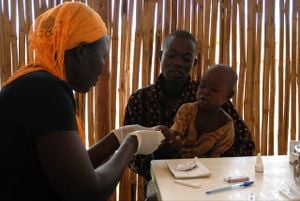
Problem being addressed[edit | edit source]
When this device was created (in 2001) microscopic diagnosis was the preferred malaria diagnosis method, but the cost of equipment and the training required were prohibitive. Previous rapid detection devices were too expensive for the developing world. This device was built for developing countries where fast diagnosis and ease of use are essential.
Detailed description of the solution[edit | edit source]
The Paracheck Pf Rapid Test device comes in two forms, a dipstick and a flat-lying device. A drop of blood is collected from a finger prick, the blood flows through a membrane after placement in a buffer, and passes over a colored anti Pf HRP-2 antisera-colloidal gold conjugate. As the test progresses, the conjugate reacts with the HRP-2 and forms a pink line. The pink indicates a positive test, and a lack of bands indicates a negative test.
Manufacturer[edit | edit source]
- Made by Orchid Biomedical Systems.
- The company's headquarters are in Goa, India.
When and where it was tested/implemented[edit | edit source]
Funding Source[edit | edit source]
References[edit | edit source]
Peer-reviewed publication[edit | edit source]
- Comparison of the Paracheck-Pf test with microscopy, for the confirmation of Plasmodium falciparum malaria in Tanzania
- Short communication: Paracheck‐Pf®: a new, inexpensive and reliable rapid test for P. falciparum malaria
Other internally generated reports[edit | edit source]
Externally generated reports[edit | edit source]
- Test of accuracy - Malaria Journal
- Assessment of two malaria rapid diagnostic tests in children under five years of age, with follow-up of false-positive pLDH test results, in a hyperendemic falciparum malaria area, Sierra Leone
- USEFULNESS OF A RAPID ON-SITE PLASMODIUM FALCIPARUM DIAGNOSIS (PARACHECK PF) IN FOREST MIGRANTS AND AMONG THE INDIGENOUS POPULATION AT THE SITE OF THEIR OCCUPATIONAL ACTIVITIES IN CENTRAL INDIA
- EVALUATION OF A RAPID DIAGNOSTIC TEST FOR ASSESSING THE BURDEN OF MALARIA AT DELIVERY IN INDIA
IP and copyright[edit | edit source]
Approval by regulatory bodies or standards boards[edit | edit source]
- ISO 9001
- CE certified
- Currently attaining FDA approval - http://clinicaltrials.gov/ct2/show/NCT00565071