Chemical fertilisers are fertilisers that are made using a chemical process. This page explains the production of the different chemical fertilisers in detail.
Ammonia[edit | edit source]
History[edit | edit source]
Around 1977, a researcher noted the possibility of using titanium oxide to make ammonia in chemical abstracts, and then went to a discussion of the whole atmospheric circulation. One of the mysteries of the atmosphere was that it had an excess of ammonia, which they have never accounted for. When the researcher considered the amount of dunes and deserts in the world, he said, "This is it!" Where do we get titanic oxide from? Sands. So he calculated it. Three acres of desert under this system would supply as much as a commercial fertilizer plant. A while later, Mollison took over the idea and made his "Mollison's third world endless nitrogen fertilizer supply system".
Requirements[edit | edit source]
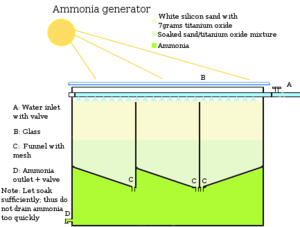
You will need a sand box, with a trickle-in system of water, and a couple of sub-surface barriers to make the water flow about. Titanium oxide can be bought by the pound if you want to. For our village, I only needed 1 bottle of it. It is available everywhere as it's a common filler in white paint, after they got rid of lead. In deserts, it can be found a plenty, because when it occasionally rains it carries it down and the nitrogen evaporates into the atmosphere.
This is also why deserts grow plants. That's why we can start a system in a desert without necessarily starting off with nitrogen fixing plants.
Construction[edit | edit source]
Fill the box with white sand and about a quarter ounce of titanium oxide.
Workings and use[edit | edit source]
Titanium oxide catalyzes atmospheric nitrogen into ammonia, endlessly,[verification needed] in the presence of sunlight. As it is catalytic, no sand or titanium oxide are used up in this reaction. Ammonia is a highly water soluble substance. Water is added and you run this endless ammonia solution off to fertilise the crops. You don't run it continuously however, because you don't want an algae buildup in the sand. Instead, you just run it cyclically; flushing out the ammonia again and again with water.
If you have a situation where you want to plant crops in sand dunes, use a pound or two of titanium oxide. You will quickly be able to establish plants in the sand, because nitrogen is continually produced after a rain. The ammonia is then is carried down into the sand. If you are going to lay down a wind barrier, this is how you do it.[1]
The titanium oxide is a catalyst, so in theory it's endless. Of course, it depends whether there's any significant washout - which in turn depends how well it's designed, built and/or operated. E.g. if the design is a bit poor but it's operated carefully, it may last many years (?); if the design is then again very good at giving space for the powder to settle and preventing it from running off, then even with careless use it may last a long time.
Struvite[edit | edit source]
Besides immediate application of urea to the soil,urine can also be improved by converting it to struvite. This can for example be done with human urine as demonstrated by the Dutch firm GMB.[2] The conversion is done by adding magnesium to the urine. An added economical advantage of using urine as fertiliser is that it contains a large amount of phosphorus, a mineral of which the production (trough mining) is rapidly decreasing as the mines are running dry.