Problem being addressed[edit | edit source]
Sepsis arises when the body's response to an infection damages its own tissues and organs. In low-resource settings, it accounts for 60-80% of all deaths. Due to underspecified symptoms, subtle symptoms, and rapid deterioration of tissue, sepsis is often diagnosed too late, and therefore treated too late.
Detailed description of the solution[edit | edit source]
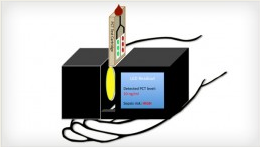
This portable, non-perishable device uses fluorescence to rapidly detect a key early biomarker for sepsis, procalcitonin (PCT). Its sensors will be cheap and it will be easy to use by health care workers and parents at home. Because sepsis mortality is related to the time of diagnosis, the device will be able to detect sepsis early enough to distinguish it from ordinary bacterial infections, thus allowing for treatment, accordingly.
Designed by[edit | edit source]
- Designed by: Lucerna, Inc, lead by Karen Wu
- Manufacturer (if different):
- Manufacturer location: New York, NY USA
When and where it was tested/implemented[edit | edit source]
Funding Source[edit | edit source]
Recipient of Grand Challenges Grant from Bill and Melinda Gates Foundation.
References[edit | edit source]
Peer-reviewed publication[edit | edit source]
Other internally generated reports[edit | edit source]
Externally generated reports[edit | edit source]
Saving Lives At Birth. (2011). Simple diagnositc device for early detection of neonatal sepsis. Retrieved December 1, 2013 from here.
Early sepsis diagnosis. Retrieved December 1, 2013 from here.