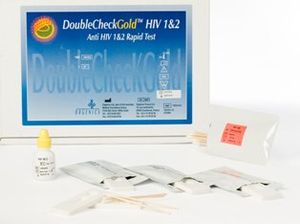
Problem being addressed[edit | edit source]
HIV/AIDS is an extreme health threat in much of the developing world. To diagnose HIV, blood samples are often required to be sent to a central laboratory to be processed, which is time-consuming and costly.
Detailed description of the solution[edit | edit source]
This small and portable immunochromatography assay allows for rapid testing of HIV-1 and HIV-2 antibodies. This assay can use whole blood, plasma, or human serum to test for HIV antibodies. The fluid is put in through the lateral flow assay and results can be achieved within 15 minutes with 99.9% sensitivity and 99.1% specificity. Antibodies specific to HIV-1 or HIV-2 proteins will react with the colloidal gold conjugate particles, enabling rapid diagnosis.
Designed by[edit | edit source]
- Designed by: ORGENICS Ltd., Israel, P.O. Box 360, Yavne, Israel 70650. Official website here.
Funding Source[edit | edit source]
The Clinton Foundation
When and where it was tested/implemented[edit | edit source]
This product was part of a multicentric study in France, Senegal, and Guinea. Tests were carried out on 2494 samples including 1720 sera, 492 plasmas and 282 whole blood to determine sensitivity and specificity of the tests with this device.