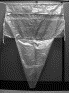
Problem being addressed[edit | edit source]
Postpartum hemorrhaging is one of the most severe causes of maternal mortality. PPH (Postpartum hemorrhaging) rates in the developed worlds have severely decreased, but they are still a great threat in developing countries. The World Health Organization suggests that there are over 100,000 deaths due to postpartum hemorrhaging every year.
Detailed description of the solution[edit | edit source]
This device is classified as one which helps in PPH estimation. This single use blood collection drape consists of a funneled and calibrated collection pouch that is attached to a plastic sheet, which is placed under the woman's buttocks immediately after delivery, tied to her abdomen with two belts located in the upper end of the drape, to estimate the postpartum blood loss.
Designed by[edit | edit source]
- Designed by:
A group of designers including Patel.
When and where it was tested/implemented[edit | edit source]
It was tested in 2003 at the District Hospital in Belgaum, Karnataka, India.
Funding Source[edit | edit source]
Global Network UMKC/ JNMC/UIC
References[edit | edit source]
Peer-reviewed publication[edit | edit source]
Patel, A., et al., Drape estimation vs. visual assessment for estimating postpartum hemorrhage. International Journal of Gynecology and Obstetrics, 2006. 93(3): p. 220-224.