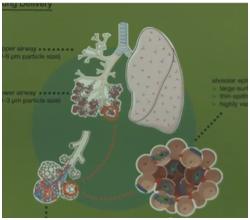
Problem being addressed
Postpartum hemorrhage is the leading cause of maternal mortality across the world. Although there is a safe and effective drug, oxytocin, that is widely administered to prevent excessive bleeding, the method of administration is often risky in resource-poor settings. Currently, oxytocin can only be administered via injection into the bloodstream by trained medical personnel, and furthermore, the drug needs to be refrigerated to retain its effects. Thus, administration of oxytocin requires sterile equipment, clean water, skilled practitioners, and electricity, all of which are not easily feasible in many developing countries.
Detailed description of the solution[edit | edit source]
Researchers have developed a more stable, low-cost, aerosol formulation of oxytocin that can be inhaled through a basic, disposable device and does not require cold-chain storage. It is a less invasive, and thus safer and simpler form of administration that would be more feasible in resource-poor settings. This delivery system does not require sterile conditions or trained medical personnel and would eliminate the risk of needle-stick injuries and reduce the possibility of transmission of blood borne diseases. The costs associated with electricity for refrigerators, proper transportation, and disposal of sharps and biohazard waste materials would also be minimized.
Designed by[edit | edit source]
- Designed by: The aerosol oxytocin delivery system is being developed by researchers led by Dr. Michelle McIntosh at Monash University in Clayton, Australia.
When and where it was tested/implemented[edit | edit source]
The product is being tested in India and Africa.
Funding Source[edit | edit source]
This project received funding from the Grand Challenge for Development Award sponsored by the US Agency for International Development (USAID), the Government of Norway, the Bill & Melinda Gates Foundation, Grand Challenges Canada and The World Bank.
References[edit | edit source]
Peer-reviewed publication[edit | edit source]
Bischof, R. J., Ibrahim, J. Parkington, H. Nassta, G., Nguyen, T., Nguyen, G., Russell, A.S., Meeusen, E.N., Morton, D. A. V. Prankerd, R. J., and McIntosh, M. P. (2012). Airway Delivery Of Oxytocin Induces Uterine Contractions In A Sheep Model: A New Approach For The Treatment Of Postpartum Haemorrhage. American Journal of Respiratory and Critical Care Medicine,185. A5619.
Other internally generated reports[edit | edit source]
Saving lives at birth: a grand challenge. (2011, July 22). Monash University. Retrieved October 15, 2012. Link available here.
New drug dose to reduce maternal deaths. (2011, March 1). Monash University. Retrieved October 15, 2012. Link available here.
2012 Victorian College of Pharmacy Foundation annual appeal. (2012, July 11). Monash University Pharmacy and Pharmaceutical Sciences. Retrieved October 15, 2012. Link available here.
Externally generated reports[edit | edit source]
Dry Powder Oxytocin. (n.d.) MANDATE: Maternal and Neonatal Directed Assessment of Technology. Retrieved October 15, 2012. Link available here.
Low cost, needle-free and non-refrigerated treatment for PPH. (2011, May 16). Saving Lives at Birth: A Grand Challenge for Development. Retrieved October 15, 2012. Link available here.
New treatment to stop mothers bleeding to death in childbirth. (2011, July 30). ABC News. Retrieved October 15, 2012. Link available here.
Saving Lives at birth: Low cost, needle-free and non-refrigerated treatment for PPH [video clip] (Aug 3, 2011). Saving Lives at Birth. Youtube video found here.