Introduction
Gallium arsenide (GaAs) is a semiconductor material that is used in a wide variety of applications ranging from circuits to solar cells. Solar cell of GaAs can be produced using both bulk and thin film growth methods.
Material Processing
Bulk Growth
There are two common ways to produce GaAs using bulk growth, Liquid-Encapsulated Czochralski (LEC) growth, and Vertical Gradient Freeze (VGF) technology. [1]
LEC growth is accomplished by melting high-purity arsenic and gallium in a high temperature vessel, and slowly cooling to produce a single crystal. The GaAs crystal produced using this method however has some impurities such as significant levels of carbon, and numerous dislocations. These impurities cause the semiconductor to be unusable for some applications.
VGF growth works by placing high purity arsenic and gallium in an enclosed quartz ampoule with a crystal of GaAs. The arsenic and gallium are melted, and then brought into contact with the GaAs crystal. When cooled slowly, a single crystal of GaAs is formed. The single crystal formed has many of the same impurities as LEC growth crystals, which restricts the utility of the crystals.
Typical dimensions of the semiconductor crystals are 1-6 inches in diameter and 2-30 inches long. The usual rate of crystal growth is 1-5mm per hour. [2]
Cutting, Polishing, and Etching
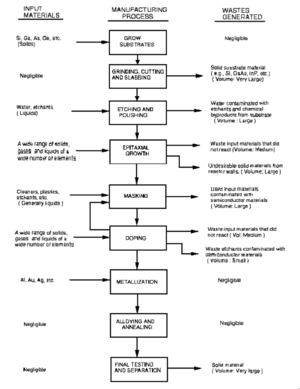
The bulk crystals (boule) has is generally cylindrical with conical ends, which are usually cut off and wasted because their diameter isn't big enough to be used. Next, the boule is ground to ensure a uniform diameter over the length of the boule which is usually done with a lathe, or similar tool. The crystals need to be aligned using x-ray diffraction and then the boule is cut into wafers. In this process nearly one third of the total mass can be lost due to the width of the cutting mechanism. [2] The sawing also leaves a damaged layer of semiconductor which is etched off. The final step in processing the semiconductor is polishing. There are two different types of waste that are produced during polishing, liquid and solid wastes. The wastewater from GaAs polishing can contain arsenic and gallium waste in concentrations between 200 and 400 ppm. [2]
Thin Film Growth
Thin films of GaAs have many advantages over large single crystals of GaAs when it comes to being used in solar cells. Thin films lack some of the impurities found in large crystals, and are capable of being used without requiring extensive slicing. The rest of the case study will be dedicated to thin film GaAs semiconductors.
The most common thin film growth methods for producing GaAs films are Vapour Phase Epitaxy (VPE), Metalorganic Chemical Vapour Deposition (MOCVD), and Molecular Beam Epitaxy (MBE).
Molecular Beam Epitaxy (MBE)
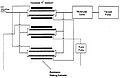
MBE is the process of depositing epitaxial films from molecular or atomic beams on a heated substrate under ultrahigh vacuum (UHV) conditions. The beams are thermally generated from elemental feedstocks in Knudsen-type effusion cells. The thermal beams travel in rectilinear paths to the substrate where they condense and grow under kinetically controlled growth conditions. [2]
Machinery Needed
Every step of the process needs a different type of machine or tool to complete it. An example of some of the equipment for each process are: Lithography - cannon i5 i-line Stepper, Gate Metal Deposition - Applied Materials Endura Sputtering System, Gate Metal Etch - Lam TCP Plasma Poly Etcher, Anneal Cap - Applied Materials Centura PECVD Nitride, Inter-Metal Dielectrics - Applied Materials Centura PECVD Oxide. [3]
Plausibility of Recycling
Size of Panels = 10 inches x 9 inches[4]
Number of Cells Per Panel = 18
Area of Cell = (90 in2/18)*.00064516 m2/in2 [((Area Panel)/(Number Cells))*(conversion from in.2 to m2)]
Area of Cell = .0032258 m2
Amount of Material per Cell = 10 grams[4]
Amount of Material per Area = 10 g /.0032258 m2 [(Material/Cell)/(Area Cell)]
Amount of Material per Area = 3100 g/m2
Peak Power per Cell = 880mW [4]
Peak Power per Area = 880mW / .0032258 m2 [(Power/Cell)/(Area Cell)]
Peak Power per Area = 272.8 W/m2
Amount of Material per Wattpeak = 3100 g/m3 / 272.8 W/m2 [(Material/Meter)/(Power/Area)]
Amount of Material per Wattpeak = 11.4 g/Wpeak
Metalorganic Chemical Vapour Deposition (MOCVD)
Average Material Utilization Efficiency = 30%[5]
Waste Material Rate MOCVD per Area = 3100 g/m2 * (1-.3) [(Material/Area)*(Percentage Waste)]
Waste Material Rate MOCVD per Area = 2170 g/m2
Waste Material Rate MOCVD per Wattpeak = 11.4 g/Wpeak * (1-.3) [(Material/Wattpeak)*(Percentage Waste)]
Waste Material Rate MOCVD per Wattpeak = 7.98 g/Wpeak
Molecular Beam Epitaxy
Material utilization efficiency for Ga = 40-70%[5]
Average Material utilization efficiency for Ga = 55%
Material utilization efficiency for As = 10-20%[5]
Average Material utilization efficiency for As = 15%
Waste Material Rate MBE = 2015 g/m2 and 7.41 g/Wpeak
Amount of waste
Approximately 50% of the weight of the material can be wasted during the manufacturing process. Of that 50%, only about 50% is then used to try and recover some of the Ga. [2]
Recycling
Collecting Waste
Semiconductor waste from MBE and MOCVD occurs in two primary methods, coating of the reactor walls and parts, and exhaust vapors drawn off of the epitaxial reactors.
The coating of the reactor walls and parts forms a solid waste material, which can be collected by simply scraping it off off reactor components. The solid waste material collected in this manner may be contaminated with dopants such as Si, Zn, C and Cr. These dopants however usually only have concentration levels of only 1018[2] atoms/cc.
The collection of semiconductor waste from exhaust vapors is not as straight forward as the collection of solid waste material. In order to collect the waste from the exhaust, the components
Process of recycling and re-purification
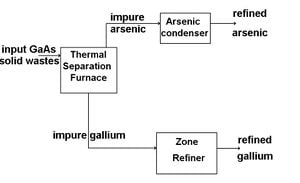
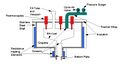
The recycling of GaAs is a relatively new idea and the current methods are deemed "satisfactory". The recovery process of Gallium is also practiced more than that of Arsenic, likely due to the toxicity of Arsenic gas. The issue with the common thermal processing of gallium is that it develops gallium and arsenic oxides, thus increasing the number of steps required to obtain a purified amount of recycled gallium.[2] The prototype process pictured is one that could be operated for a relatively low cost and return the recovered materials directly back into the crystal growing process. This process consists of only three steps and three pieces of equipment, a thermal separation furnace, arsenic condenser (for purification), and a zone refiner (for gallium purification).
-
Thermal Separation Furnace
-
Purification of Ga
-
Purification of As
The idea of down cycling GaAs waste has not been sufficiently examined yet. The industry using GaAs is just around 20 years old and any environmental effects of the waste material have yet to be realized causing a delay in the recycling process.[2] Currently, GaAs is only recycled (or subject to testing of recycling methods) to reduce the cost of manufacturing by improving the percentage of GaAs recovered and reused. However, if a recycling method were to be streamlined and implemented into the manufacturing process companies could see great yields in recycling. Since about 50% of the GaAs is lost during the manufacturing, if GaAs waste could be recycled to be directly implemented back into the manufacturing process, theoretically you could get twice the amount of semiconductor as you do today. This, of course, may not be a realistic goal due to the fact that recycling may not yield 100% return, and other methods of recycling may be found to be more cost effective, faster, or easier for a company to do without disturbing the actual manufacturing process.
Safety
References
- ↑ R.L. Adams, Growth of high purity GaAs using low-pressure vapour-phase epitaxy, Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment, Volume 395, Issue 1, 1 August 1997, Pages 125-128, ISSN 0168-9002, 10.1016/S0168-9002(97)00624-4. (http://www.sciencedirect.com/science/article/pii/S0168900297006244) Keywords: Low-pressure vapour-phase epitaxy; LPVPE; GaAs
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 Swartzbaugh Joseph, Sturgill Jeffery. Reduction of Arsenic Wastes in the Semiconductor Industry, 1998
- ↑ Mikkelson James, Tomasetta Louis. August 2003 High-Density GaAs Integrated Circuit Manufacturing
- ↑ 4.0 4.1 4.2 GaAs Solar Panel; Product Data Sheet; 3554 Chain Bridge Road, Suite 103, Fairfax, VA 22030, http://www.spacequest.com/products/SP-X.pdf , (accessed September 2011).
- ↑ 5.0 5.1 5.2 V.M. Fthenakis, B. Bowerman, Environmental Health and Safety (EHS) Issues in III-V Solar Cell Manufacturing, National PV EHS Assistance Center (http://www.bnl.gov/pv/files/pdf/art_168.pdf)