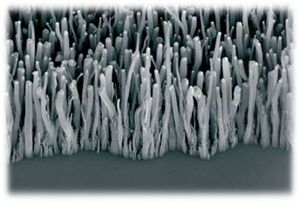
A review of current and prospective methods for efficient large-scale production of carbon nanotubes
Since carbon nanotubesW were first observed in 1991, years of research have been invested into mass producing these remarkable structures. Carbon nanotubes (CNTs) have the potential to revolutionize materials engineering, making it possible to create materials with novel properties. The optimization of CNT synthesis processes on a large scale is necessary to make nanotubes a feasible material for implementation in nano-scale devices and materials.
Overview[edit | edit source]
CNTs are a highly desirable material because of their unique properties. They are a derivative of fullerenesW, formed from chains of C2 dimersW and C3 trimersW with hemispherical caps.[1] Experiments have shown that, with a potential tensile strengthW of ~25 GPa and Young's ModulusW as high as 1 TPa, it is one of the strongest and stiffest materials available. Carbon nanotubes are also highly conductive and have unique optical properties.[2] They have potential as low-loss connectors in nanoscale integrated circuitsW and field-effect transistorsW, photovoltaic cellsW, in composite materialsW, and more. Carbon nanotubes are divided into two main categories: Single-Walled Nanotubes (SWNTs) consisting of a single layer of carbon, and Multi-Walled Nanotubes (MWNTs) made up of several nested tubes.
Common Synthesis Techniques[edit | edit source]
Arc Discharge and Laser Ablation[edit | edit source]
The first method that was successfully used to synthesize CNTs in small quantities was the arc discharge method. Opposing anode and cathode terminals made of 6-mm and 9-mm graphite rods respectively are placed in an inert environment (He or Ar at ~500 Torr). A strong current, typically around 100 A (DC or AC), is passed between the terminals generating arc-induced plasma that evaporates the carbon atoms in the graphite. The nanotubes grow from the surface of these terminals.[3] A catalyst can be introduced into the graphite terminal. Although MWNTs can be formed without a catalyst, it has been found that SWNTs can only be formed with the use of a metal catalyst such as iron or cobalt.[4] A process called Laser Ablation, first developed in 1995, uses a similar principle to produce nanotubes. Carbon is evaporated at high temperatures from a graphite target using a powerful and focused laser beam. In the most basic laser ablation technique, a 1.25-cm diameter graphite target is placed in a 2.5-cm diameter, 50-cm long quartz tube in a furnace controlled at 1200oC and filled with 99.99% pure argon to a pressure of 500 Torr. A pulsed Nd:Yag laserW beam at 250mJ (10 Hz) is focused using a circular lens and the beam is swept uniformly across the graphite target surface. The nanotubes, mixed with undesired amorphous carbon, are collected on a cooled substrate at the end of the chamber.[5]
Both of these methods have limited potential for scale-up. Solid graphite must be evaporated at >3000°C to source the carbon needed, the nanotubes produced are in an entangled form, and extensive purification is required to remove the amorphous carbon and fullerenes that are naturally produced in the process.[6]
Chemical Vapor Deposition[edit | edit source]
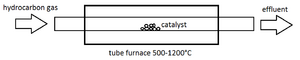
Chemical Vapor Deposition (CVD) has the highest potential for mass production of carbon nanotubes. It is a versatile method that can produce bulk amounts of defect-free CNTs at relatively low temperatures.
The generalized process for producing CNTs using CVD is outlined here. A substrate material (e.g. alumina, quartz), is cleaned in preparation for the catalyst deposition. A porous substrate may be desired, so electrochemical etching with a hydrofluoric acid/methanol solution may be performed. Nanotubes can grow at a higher rate on a porous substrate, suggesting that carbon can diffuse through the porous substrate layer and feed growing nanotubes.[7] A catalyst (e.g. iron, nickel) is deposited onto the substrate by thermal evaporation. The furnace is raised to a temperature between 500-1200°C and a hydrocarbon gas such as acetylene, ethylene, or carbon monoxide is slowly pumped into the reactor[8] (Figure 1). At these high temperatures carbon dissociates from the feedstock molecules and diffuses onto the catalyst. The atoms arrange themselves into a sheet of nanotubes on the substrate, combined with impurities such as amorphous carbon, fullerenes, as well as the catalyst material. In most cases these impurities must be removed using a purification step. An acid treatment followed by sonificationW is popular. The specifics of these processes are described below.
Growth Mechanisms

The growth mechanisms of carbon nanotubes is not fully understood. Many different models of carbon nanotube growth have been proposed, and the most accepted model which is consistent with solid-phase growth is described here. Small carbon atoms diffuse around a large metal catalyst atom which is assumed to have a spherical shape. The carbon nucleates on the sides of the metal, forming a tube structure (Figure 2). The location of the catalyst material during formation depends on the strength of the interatomic forces between the metal atoms and the substrate: if the metal is weakly bonded to the substrate it may remain in place as a nanotube forms vertically from the substrate (this mechanism is entitled base growth); otherwise the metal may be carried at the tip as the tube grows beneath (entitled tip growth).[1]
Catalyst
The catalyst provides a base around which the nanotubes can grow and the material chosen for CVD synthesis can wildly vary the yield and quality of CNTs produced. Conventionally, the catalysts are transition-metal nanoparticles (usually iron, nickel, molybdenum) deposited on a support material such as alumina or silica. However vaporized catalysts have also been used such that the nanotubes form in the gaseous phase and are condensed on cold surfaces.[4]
The catalyst becomes deactivated when it is coated by undesirable amorphous carbon, stopping the CNT growth process. An ideal catalyst material should have large surface area and pore volume (properties that must not sinter out at high temperatures), as well as strong metal-support interactions.[6]
The average diameter of the carbon nanotubes produced by CVD is directly related to the size of the catalyst particles. This relationship can be illustrated by the growth mechanisms shown in Figure 2. As the nanotubes tend to grow perpendicular to the surface of the catalyst, the diameter of the particle dictates the diameter of the nanotube.[8] Thus CVD provides direct control over the size of carbon nanotubes, making it a viable option for materials processing.
Carbon Feedstock
In CVD the carbon is supplied by a hydrocarbon gas. The reactivity of the molecule is an important factor determining which hydrocarbon to use. The reactivity is dictated by the strength of the covalent bonds between the carbon and the hydrogen atoms - that is, a more reactive molecule requires less energy to decompose into its components. This decomposition is achieved by elevating the temperature, providing the molecule with kinetic energy to help break the bonds. Thus, ethylene (C2H4) and acetylene (C2H2) are usually chosen over methane and carbon monoxide as the carbon feedstock because of their higher reactivity. Benzene, methanol, and ethanol have also been successfully used for various experiments.[4]
Purification
The carbon nanotubes can be separated from impurities and catalyst particles using a variety of techniques. The most common post-production steps are (1) dispersion, followed by (2) acid reflux and finally (3) micro-filtration. Dispersion is performed by sonication of the nanotubes with a surfactant, such as Triton X100 (a type of detergent). A solution of 0.01ml of Triton X100 and 0.08 litres of water per gram of carbon is used to separate the nanotubes from the impurities. The acid reflux treatment is often done using 0.4 litres of nitric acid per gram of dispersion product over 10 hours. This step is slow, and therefore requires several processing lines to maintain productivity. Filtration is performed through a polytetrafluoroethylene (PTFE) membrane over approximately 20 minutes, but may require many runs to achieve the desired purity. On average, about 90% of the nanotube yield can be recovered after the purification process.[9]
Ordered Nanotubes by CVD[edit | edit source]

Recent advances have shown that nanotubes can be synthesized in an ordered manner. Catalysts can be deposited onto a porous silicon substrate in a pattern through a shadow maskW so that nanotubes can only nucleate at specific sites. The result is known as a 'nanotube forest' because the nanotubes grow vertically from the catalyst sites, with each tube held up by van der Waals forcesW from surrounding tubes. Directed growth of SWNTs has also been demonstrated. Catalyst material was printed onto the tops of silicon pillars selectively, and after CVD, SWNTs were shown to grow suspended from pillar to pillar, as shown in Figure 3.[6] The growth of carbon nanotubes in a controlled way is an essential skill to master in order to effectively produce CNT-based nanotechnologies.
Optimization of CVD Synthesis[edit | edit source]
As CVD shows the most promise for mass production of carbon nanotubes, extensive research has been done to improve the yield and energy efficiency of the synthesis process. Manufacturing efficiency can be quantified in a very simple way:
The value added time encompasses all the time in a process that is put into creating a marketable product. To increase the efficiency either time must be used more productively, or the process must be completed in less time overall. In addition, increasing the yield of useful nanotubes increases the efficiency as the same amount of time adds more value to the manufacturing process. A more efficient process also produces less waste, as time must be spent consolidating the waste material post-production.
By extension the overall cost of the synthesis process is decreased with increasing efficiency. The ultimate goal is to minimize the cost so that nanotubes can be used in a variety of applications. A few of the most promising methods developed to achieve this goal are highlighted below.
Water Assisted CVD ("Supergrowth")[edit | edit source]
A catalyst is limited in its ability to nucleate CNTs. The reason that the catalysts become deactivated is that amorphous carbon naturally forms around the catalyst over time, smothering the available nucleation sites. Models of catalyst decay have shown strong correlation to models of radioactive decayW. Therefore the height of the forest H(t) under set growth conditions can be described by exponential decay,
where β is the initial growth rate, τ is the catalyst lifetime, and βτ intuitively gives the maximum height of the forest. Adding a small amount of water vapor (in the ppm range) to the ambient atmosphere can increase the lifetime of the catalyst material. Remarkably this exponential decay model has predicted catalyst behaviour very precisely. The catalyst lifetime, and therefore the height of the forest, can be maximized at a specific water-ethylene ratio. Recent experiments even show that this optimum ratio is independent of growth time and temperature, which points to the fact that water is changing a fundamental aspect of the carbon nanotube growth process.[7]

Although the effect of water is poorly understood, it has been hypothesized that water etches amorphous carbon without etching the nanotubes. The effect is that nucleation sites are kept free of amorphous carbon so that the catalysts are not inactivated while nanotubes are unaffected, fostering increased growth (Figure 4). Supergrowth has generated super-pure (99.98% purity) vertically aligned SWNT forests in the millimeter range – this is an unprecedented achievement in ordered CNT synthesis.[11]
CVD with No Catalyst[edit | edit source]
Catalyst materials can encourage nanotube growth; however they must be purified out at the end of the synthesis process. It has been shown that, remarkably, an external catalyst is not necessarily needed to produce carbon nanotubes. This eliminates the need for purification and thus increases the efficiency of the process. A simple method has been developed by Fadour et al. to grow CNTs on etched stainless steel. At an operating temperature of about 700-850oC this method shows promise for increasing synthesis efficiency by eliminating the need for a pretreated catalyst and by depositing nanotubes directly onto a steel substrate. The process is performed as follows: substrate cleaning for 30 minutes with acetone in an ultrasonic bath, substrate etching in 35-38% hydrochloric acid, substrate heat treatment at 850oC, acetylene injection at 700oC and hold in nitrogen gas, followed by cool down.[12]
Hot-Filament CVD[edit | edit source]
One of the main sources of inefficiency in any processing system is the need to sustain elevated temperatures for synthesis, so reducing the operating temperature of the CVD process would greatly enhance energy efficiency. Chemical Vapor Deposition has been demonstrated at low temperatures (~550oC) with the assistance of a heated filament (a process termed HF-CVD). A filament is pre-heated in a near-vacuum atmosphere to about 2000oC, and is used to heat the inflowing gases in the CVD chamber. The addition of this filament enables synthesis of highly pure nanotubes, but increasing the temperature above 750oC was shown to result in the deposition of undesired amorphous carbon. This alteration to the conventional CVD process has the potential to greatly enhance synthesis efficiency.[13]
Plasma Enhanced CVD[edit | edit source]
The nucleation rate of carbon nanotubes can be dramatically increased by ionizing the incoming gases into plasmaW phase. Plasma-Enhanced Chemical Vapor Deposition (PECVD) allows carbon to diffuse more readily and increases the yield of nanotubes. The plasma can be generated by direct current across two terminals, or more commonly by exciting the gas through wire coils with a high frequency current (13.56 MHz is the standard in industry). Although strong currents are required to generate the plasma, it has been shown that the operating temperature in the furnace can be reduced by half or more. The same experiment showed that applying an electric field causes an alignment among the growing nanotubes and the strength of the field is proportional to the degree of alignment. In addition, when NH3 is pumped in with the hydrocarbon gas, the rate of nanotube nucleation is increased. This is due to the fact that NH3 etches amorphous carbon most strongly at bias voltages above -300V. These results are promising as they provide a well-established method for increasing synthesis efficiency by producing higher yields of aligned CNTs at relatively low temperatures.[14]
Applications[edit | edit source]
There is no doubt that carbon nanotubes have far-reaching macroscopic and microscopic applications. As synthesis methods are being improved, extensive research is being done to one day implement CNT technologies. A few interesting applications are discussed here.
SWNT-polymer solar cells
Single-walled nanotubes have a promising future in the photovoltaics industry, bringing us closer to sustainable and efficient solar energy systems. It has been demonstrated that utilizing a composite of P3OT polymer and SWNTs could help improve the efficiency of third-generation thin film solar cells. The allure of nanotubes is that, because of their high length-diameter ratio, they provide an excellent pathway for the diffusion of excitons in the conducting polymer. This improves charge transfer and carrier mobility in the cell when the polymer is doped with a small amount of SWNTs. In addition, the structural and thermal properties unique to CNTs can be exploited. Using a nanotube-polymer composite can greatly increase the tensile strength and Young's Modulus of solar cells. The high thermal conductivity of nanotubes is also an attractive quality. These properties are very important to consider when constructing large photovoltaic farms, so carbon nanotubes could become an integral part of next-generation solar cells[15]
CNTs in Nanoelectronics
As electronic structures become smaller, it is clear that current materials will soon be too inefficient for use in nano-scale circuits. For example, copper is now the material of choice for interconnects in integrated circuits – however they have high resistivity at small scales and low current density. Carbon nanotubes are being researched as a possible replacement for copper as interconnects in integrated circuits due to their high current-carrying capacity, and stable structural and thermal properties.[16]
Futhermore, carbon nanotubes have been used to replace silicon as the semi-conducting channel in field-effect transistors, creating so-called CNT-FETs. A single SWNT is used to connect the two main electrodes, offering improvements in electron transfer and device stability. As transistors form the basis of every day electronics, CNTs have the potential to increase data transfer speeds and allow the design of nano-scale electronic components.[17]
Current Challenges and Limitations to Commercial Implementation of CNT Technology[edit | edit source]

At present the mechanisms of carbon nanotube growth are not fully understood and therefore it has been difficult to manipulate the synthesis of these microstructures. In fact, synthesis processes thus far have only been optimized by trial and error. Research is being conducted to understand these growth mechanisms, but so far the demand for carbon nanotube technology has outpaced our ability to fully grasp the processes involved in producing them.
Another roadblock is that carbon nanotubes, although they have great potential for many applications, have not been produced in large enough quantities and at low enough cost to replace any currently available materials in the market. According to a recent analysis, the bulk cost to produce SWNTs is about $1706 per gram. A large part of this cost (approximately 80%) comes from the synthesis step alone (Figure 5). Clearly, improvements in the efficiency of the synthesis process must be implemented before CNTs can become a viable technology.[9] Although MWNTs are less costly to synthesize, tonnage quantities of high purity and high quality nanotubes have yet to be produced. Despite the current roadblocks, research is bringing the widespread implementation of carbon nanotube technology closer to reality.
References[edit | edit source]
- ↑ 1.0 1.1 1.2 Charlier, J-C, and Sumio Iijima. Growth Mechanisms of Carbon Nanotubes. In M.S Dresselhaus, G Dresselhaus, Ph. Avouris (Eds.) Carbon Nanotubes: Synthesis, Structure, Properties, and Applications. (pp.55-79). New York: Springer, 2001.
- ↑ Seo, J.W, and L. Forró. Structural properties of nanoelectromechanical systems applications. In S. Saito and A. Zettl (Eds.) Carbon Nanotubes: Quantum Cylinders of Graphene. (pp. 135-70). New York: Elsevier, 2008.
- ↑ Ebbesen, T.W. Large-scale synthesis of carbon nanotubes.Nature (1992) 358, 220 - 222
- ↑ 4.0 4.1 4.2 Mann, David. Synthesis of carbon nanotubes. In M.J O'Connell (Ed.) Carbon Nanotubes: Properties and Applications (pp. 19-50). Menlo Park, Calif.:Taylor and Francis, 2006
- ↑ Guo T. et al. Self Assembly of Tubular Fullerenes. J. Phys. Chem. 1995, 99, 10694-97
- ↑ 6.0 6.1 6.2 6.3 Dai, Hongjie. Nanotube growth and characterization. In M.S Dresselhaus, G Dresselhaus, Ph. Avouris (Eds.) Carbon Nanotubes: Synthesis, Structure, Properties, and Applications. (pp.29-51). New York: Springer, 2001.
- ↑ 7.0 7.1 7.2 Joselevich, E., Dai, H. J.; Liu, J.; Hata, K.; Windle, A., Carbon Nanotube Synthesis and Organization. Top Appl Phys 2008, 111, 103-166
- ↑ 8.0 8.1 Jacques, D. et al. Synthesis of Multi-Walled Carbon Nanotubes. Center for Applied Energy Research (2003)
- ↑ 9.0 9.1 Isaacs, J.A. et al. Economic assessment of single-walled carbon nanotube processes. J Nanopart Res, Springer. (2009)
- ↑ Muir, Alastair. Calculating Process Efficiency in Transactional Projects. http://web.archive.org/web/20091217103304/http://www.isixsigma.com:80/library/content/c031208a.asp (2009)
- ↑ Futaba, D.N. et al. Kinetics of Water-Assisted Single-Walled Carbon Nanotube. Phys. Rev. L. 056104 (2005)
- ↑ Badour, C.E. et al. A simple thermal CVD method for carbon nanotube synthesis on stainless steel 304 without the addition of an external catalyst. Carbon 47 (2008), 313-347.
- ↑ Jayatissa, A.H., K. Guo. Synthesis of carbon nanotubes at low temperature by filament assisted atmospheric CVD and their field emission characteristics. Vacuum 2009, 83, 853–856
- ↑ Chhowala, M. et al. Growth Process conditions of vertically aligned carbon nanotubes using plasma enhanced chemical vapor deposition. J. Appl. Phys. 2001, 90, 5308-5317
- ↑ Landi, B. J.; Raffaelle, R.P.; Castro, S.L.; Bailey, S.G., (2005). Single-wall carbon nanotube-polymer solar cells. Progress in Photovoltaics: Research and Applications (2005) 13: 165–172
- ↑ Banerjee, K. and Srivastava, N. Are Carbon Nanotubes the Future of VLSI Interconnections? Proceedings of the 43rd annual Design Automation Conference (2006) 809-814
- ↑ Daenen, M. et al. The Wondrous World of Carbon Nanotubes:a review of current carbon nanotube technologies. (2003)
