Tertiary Treatment
Tertiary treatment of effluent provides a final stage to raise its quality before it is released into the environment. A treatment plant may use opt to use more than one tertiary treatment process, such as combining nutrient removal with filtration. If disinfection is practiced, it is always the final process. Tertoary treatment is also called advanced treatment, "effluent polishing", or in the case of the Arcata Marsh "enhancement".
Types of Treatment
Filtration After secondary treatment there are generally still some suspended solids left in the effluent. The main reason for this is that the secondary process is not perfect at filtering out microorganisms left over from the biological filtration processes. This "biological floc" is usually filtered out mechanically by using a multimedia filter that has progressively smaller and smaller filter grain sizes. This reduces the amount of back washing necessary to clean the filter.
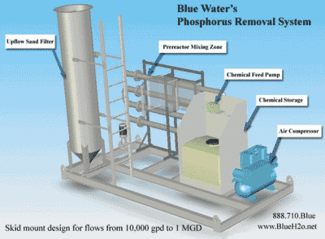
Nutrient removal Wastewater may contain high levels of nutrients such as nitrogen and phosphorus. The excessive release of these to the environment can cause the overgrowth of weeds, algae, and cyanobacteria (blue-green algae). A rapid growth in the population of algae can result is an algal bloom of a magnitude such that the numbers are unsustainable. Eventually most of the algea die. The decomposition of the algae by bacteria uses up so much oxygen in the water that most or all of the animals living in the water die, which creates more organic matter for the bacteria to decompose. In addition to causing deoxygenation, some algal species produce toxins that contaminate drinking water supplies. Different treatment processes are required to remove nitrogen and phosphorus. Phosphorous is generally removed using chemicals such as alum, or ferric chloride which precipitate to form a salt and hydronium or hydroxide ions. These ions can cause the waters pH to fluctuate. Generally lime is used to stabalize the pH of the effluent. These reactions require a settling basin to remove the precipitants. Removing nitrogen can be accomplished using either chemical or biological processes. Nitrogen is generaly present in the form of amonia. The chemical process simply raises the pH of the effluent. This converts the amonia to amonium which can be removed from teh water simply by passing large quantities of oxygen through it. The biological process simply uses activated sludge for an extended period of time, typically around 15 days.[1]
Disinfection
The purpose of disinfection in the treatment of wastewater is to substantially reduce the number of microorganisms in the water to be discharged back into the environment. The effectiveness of disinfection depends on the quality of the water being treated (e.g., cloudiness, pH, etc.), the type of disinfection being used, the disinfectant dosage (concentration and time), and other environmental variables. Cloudy water will be treated less successfully since solid matter can shield organisms, especially from ultraviolet light or if contact times are low. Generally, short contact times, low doses and high flows all militate against effective disinfection. Common methods of disinfection include ozone, chlorine, or ultraviolet light.
Chlorination remains the most common form of wastewater disinfection in North America due to its low cost and long-term history of effectiveness. One disadvantage is that chlorination of residual organic material can generate chlorinated-organic compounds that may be carcinogenic or harmful to the environment. Residual chlorine or chloramines may also be capable of chlorinating organic material in the natural aquatic environment. Further, because residual chlorine is toxic to aquatic species, the treated effluent must also be chemically dechlorinated, adding to the complexity and cost of treatment.[2]
Ultraviolet (UV) light can be used instead of chlorine, iodine, or other chemicals. Because no chemicals are used, the treated water has no adverse effect on organisms that later consume it, as may be the case with other methods. On the other hand, UV radiation causes damage to the genetic structure of bacteria, viruses, and other pathogens, making them incapable of reproduction. The key disadvantages of UV disinfection are the need for frequent lamp maintenance and replacement; and the need for a highly treated effluent to ensure that the target microorganisms are not shielded from the UV radiation (i.e., any solids present in the treated effluent may protect microorganisms from the UV light). In the United Kingdom, light is becoming the most common means of disinfection because of the concerns about the impacts of chlorine in chlorinating residual organics in the wastewater and in chlorinating organics in the receiving water. Edmonton, Alberta, Canada also uses UV light for its water treatment.[3]
Ozone Ozone is very unstable and reactive and oxidizes most organic material it comes in contact with, thereby destroying many pathogenic microorganisms. Ozone is considered to be safer than chlorine because, unlike chlorine which has to be stored on site (highly poisonous in the event of an accidental release), ozone is generated onsite as needed. Ozonation also produces fewer disinfection by-products than chlorination. A disadvantage of ozone disinfection is the high cost of the ozone generation equipment and the requirements for highly skilled operators.[4]
References
- ↑ Davis, Mackenzie L. and Susan J. Masten. Principles of Environmental Engineering and Science. New York. Mc Graw Hill. 2004.
- ↑ http://www.americanchemistry.com/s_chlorine/sec_content.asp?CID=1198&DID=4534&CTYPEID=107
- ↑ http://www.ozonia.com/tips/wastewater_uv.html
- ↑ http://www.ozonia.com/tips/wastewater_uv.html