OYSTER RESTORATION USING DESIGNED REEF STRUCTURES by Wayne Young
Overview[edit | edit source]
Oyster rehabilitation is very complex. Designed reef structures fill a niche in a much larger oyster rehabilitation need, and tend to have a supporting or secondary rather than leading role. Consultation and coordination with knowledgeable practitioners is essential. This section surveys major planning considerations and issues associated with the use of designed reef structures in support of oyster rehabilitation; it is an introduction to the topic. Practical information from previous use of Reef Balls with oyster rehabilitation objectives is also discussed. These projects were correlated or integrated with local area oyster rehabilitation programs as a best practice.
Oysters[edit | edit source]
Oysters, a keystone species of benthic filter feeders in temperate estuaries and coastal bays, are a type of primitive bivalve with global distribution. An oyster’s filtering capabilities enables it to remove large quantities of plankton, nutrients, bacteria, and sediment from the water. A healthy adult oyster can filter 25-60 plus gallons of water per day. A large, healthy oyster population can improve water quality. Oyster beds and reefs also provide valuable habitat for other benthic organisms and fish species. Predation, harvesting pressure, water quality degradation (e.g., high nutrient loads), and diseases, especially parasites in recent years, have combined to drastically reduce many oyster populations. Combined stresses can cause natural oyster populations to decease below the quantity to rebuild the stock at a rate that offsets or is greater than cumulative losses. Rehabilitation may be an option if environmental and institutional conditions are otherwise favorable. Oysters have a complex life history that makes rehabilitation extremely challenging. A basic understanding of the oyster and its environment is fundamental to rehabilitation project design. Effective use of designed reef structures must consider the life histories of the oyster species that will be used, the complexities of oyster propagation, growth and mortality factors, the potential for performance to vary significantly according to prevailing conditions, and rehabilitation issues within the ecosystem.
Designed Reef Oyster Rehabilitation Concept[edit | edit source]

Constructed beds and structures are routinely used to provide hard substrate for direct attachment by oyster larvae or to serve as a foundation for seeding oysters for rehabilitation and commercial oyster cultivation. As a matter of practical necessity or to facilitate harvesting, such structures may not be configured in the vertical or horizontal dimensions so as to approximate the layout and irregular elevations of natural oyster reefs. Use of designed reef structures in rehabilitation projects is an adaptation of these longstanding oyster cultivation practices.

Rehabilitation of oyster and coral reefs apply the same basic concept. Designed reef structures provide hard substrate for attachment by larval oysters, simulating natural structure (e.g., relic oyster shell formations). The structure provides a core for the reef. Oysters that colonize the core structure form a living veneer of marine organisms. The core and veneer, when fully developed, replicate the functional performance of a natural reef many years in age. Applying this concept, Reef Balls were used successfully as substrate for natural and hatchery-assisted “spat sets” by the American Oyster (Crassostrea virginica) in the Tampa and Chesapeake Bay areas, respectively.
Actual and potential applications of designed reef modules include: Reestablishment of vertical structure for oyster colonization.
- Use of the inner cavity as a grow-out area for spat on shell for protection from
predation by fish (e.g. cow-nosed ray).
- Deployment on oyster bars or suitable rehabilitation project sites for natural
colonization.
- Deployment on oyster bars or suitable rehabilitation project sites with spat set
directly on or manually attached (e.g. spat on shell) to the modules.
- Deployment of modules (with or without spat attached) in patterns that
approximate the configuration of a natural reef system.
- Deployment on oyster sanctuaries or oyster aquaculture sites to deter
unauthorized mechanical harvesting or protect grow-out cages.
- Deployment at corners or along sides of oyster sanctuaries or oyster aquaculture
sites to provide underwater boundaries detectable by fathometers and sonar.
- As secondary benefit for other designed reef applications (e.g., establishment of
reef habitat for fish, shoreline stabilization structures).
Rehabilitation Project Planning[edit | edit source]
Oyster rehabilitation is usually an ecosystem-wide issue that requires multi-faceted activity on a large-scale. A combination of physical alterations to the substrate of natural oyster bars, overharvesting, or disease (e.g., parasites) are likely to be present. Individual and cumulative stresses from harvesting, disease, water quality degradation, sedimentation, and other factors may have reduced the oyster population below levels that are sufficient to sustain the species or support a commercial fishery. Oyster rehabilitation project sites may also susceptible to unauthorized harvesting, although it is possible to design projects to help mitigate this threat. Rehabilitation on an ecosystem scale involves environmental, fisheries management, and regulatory issues, large costs and substantial effort. Sponsor and institutional support are essential. Smaller-scale oyster reef rehabilitation projects may be within available resources, and could be used to help development of oyster seed stock, to provide localized reef habitat, and to support secondary objectives such as reef structure beneficial to other marine species.
Scoping Projects[edit | edit source]
Planning meetings should be conducted to scope a prospective project. Understanding the biological and ecological issues, local environmental conditions, institutional concerns (including fisheries management issues), and regulatory requirements are fundamental to implementing successful projects. Identifying actual and potential stresses to oyster populations is a fundamental consideration.
To assist with identifying issues and concerns and obtaining advice and support, project proponents and planners should reach out to stakeholders and engage their participation. Scientists, natural resource agencies, and regulatory authorities are core stakeholders. Because underwater structures are involved, the process should include those who could be directly or indirectly affected by a project, such as recreational boaters, sailing groups, commercial and recreational fishermen, divers, swimmers, and environmental interest groups that include marine issues in their agendas. Potential co-sponsors also merit consideration in project planning. A large body of relevant literature is accessible through Internet searches and should be consulted.
The differing and often competing habitat requirements between species of interest to natural resources, fisheries management, and environmental stakeholders may be a constraining factor as needs of “client species” are represented. Construction of an oyster reef usually involves conversion bottom habitat from one form to another, unless a structure is placed on an existing active oyster bar. The conversion occurs even when a selected site is a former oyster bed or reef that has become buried by sediment. The conversion of benthic habitat may be a fisheries management issue, for example in the United States, where the existing bottom has been classified as “essential fish habitat” by the National Marine Fisheries Service.
The planning factors that are identified should be assessed, project objectives developed, and a consensus reached among stakeholders insofar as practicable regarding project specifics, facilitating such regulatory approvals as may be necessary. However, a consensus may be difficult to achieve inasmuch as oyster rehabilitation is often a hotly debated; there are competing interests and concerns, and in some cases, vested interest in particular rehabilitation activities. Therefore, considerable care must be exercised in scoping and developing projects. Integrating designed reef projects with established oyster rehabilitation activities or as elements of rehabilitation projects sponsored by others may help offset the potential for disagreements. However, it may be necessary to adjudicate the various concerns that associated with any rehabilitation project through the applicable regulatory process.
Setting Project Objectives[edit | edit source]
Use of designed reef structures to provide high relief substrate for oyster recruitment is typically expensive, resource intensive, and small scale relative to overall population needs. Projects should be designed to provide benefits to the environment in the event that oyster recruitment or performance is substantially below anticipated results or does not occur, for example, if reefs relying on natural propagation do not receive a spat set. In order to accomplish this, projects should include both primary and secondary objectives where practicable. Secondary objectives could include fisheries habitat to improve carrying capacity for reef fishes, recreational diving opportunity, shore stabilization, research, and deterrence of unauthorized mechanical harvesting. These could also be primary objectives for some projects, with oyster rehabilitation serving as a secondary objective.
Large underwater structures can help deter unauthorized mechanical harvesting. But inclusion of deterrence as a project objective may be controversial, if not offensive to some. Implications that poaching could potentially occur should be well validated before being included as a specific project objective. Underwater structures can also snag nets and other fishing gear that may be authorized fishing practices in or in proximity to project areas. Therefore, recreational and commercial fishing interests need to be effectively addressed through the scoping process.
Regulatory Requirements[edit | edit source]
Project planning must effectively determine the applicable regulatory requirements.Project implementation must conform to regulatory criteria that are prescribed. Among the issues that have been considered include, but are not limited to, oyster species selection (including hybrids and non-indigenous species), conversion of habitat from one species to another, risk, location, water depth (biological and navigation), pollution, physical conditions and effects, benefits, costs, and effects relative to other rehabilitation projects. The cost and effort to complete studies that may be required to satisfy regulatory issues can be substantial. Therefore, linking or correlating projects with existing oyster rehabilitation work can reduce and make practical applications that might otherwise be difficult to obtain regulatory approvals.
Biological Planning Considerations[edit | edit source]
Species Selection[edit | edit source]
A best practice is to select a species that has a proven record of performance under prevailing location conditions. However, only species that are approved for use in the project area must be used. There may be one or several indigenous species, a hybrid of an indigenous species especially bred to be disease resistant, or an introduced (non-indigenous) species. Among the oysters that are cultured are the Eastern, or American oyster, European oyster (Ostrea edulis), Pacific oyster (Crassostrea gigas), and the Suminoe, or Asian oyster (Crassostrea ariakensis). Sterile oysters of a suitable species may be beneficial to commercial aquaculture (no energy lost to reproduction) but would not normally be candidates for a rehabilitation project because they can’t reproduce. However, if a project site were located within the hydrodynamic circulation pattern of a reliable source of naturally reproducing oysters, then use of sterile oysters to “jump-start” an oyster reef development with reliance on natural propagation for future recruitment of native stock might be an option.
The oyster species that is selected must be amenable and cost-effective to propagation, transplantation, or stabilization. Oyster species often display characteristics that are evolutionary adaptations to conditions that prevail in their primary ranges. Although one species might have a characteristic that seems potentially favorable to introduction in another part of the globe, the full characteristics of the species need to be assessed to determine actual suitability.
Rigorous international protocols – the International Council for the Exploration of the Sea’s ICES 1994 Code of Practices on the Introductions and Transfers of Marine Organisms - were developed to guide the intentional introduction of non-indigenous species. These standards are based on past introductions, intentional and non-intentional, that have had adverse or disastrous consequences. There are also national laws, such as the Non-Indigenous Aquatic Nuisance Prevention and Control Act of 1990 (P.L. 101-646) in the U.S., as well as laws in several states, to prevent the introduction of non-indigenous and invasive species that could be harmful. A non-indigenous oyster species should not be used unless it has been certified and approved by proper authority. The research and risk assessments that are associated with the applicable protocols and institutional requirements are expensive, resource intensive, and time consuming. [[Figure 0 Oyster spat on shell. Photo: Wayne Young, Maryland Environmental Service. Brood stock or seed oysters, whether indigenous or non-indigenous, that are imported from another location, may need to be subjected to a quarantine to insure that the stock is not infected, and the packing material is also not infected. Once all requirements and safety precautions have been satisfied, a non-indigenous species may be an option, provided that it is a suitable species relative to project objectives. If natural reef building is a project objective, then the oyster species that is selected must be an effective reef builder. The American and Pacific oysters are strong space competitors. When conditions are favorable, each species can establish dense populations and can potentially displace other oyster species. The Suminoe oyster tends to build relatively flatter profiles and does not appear to be as good a space competitor as either the American or Pacific oyster. The American oyster tends to become dormant in unfavorable conditions, such as low dissolved oxygen. In contrast, the Suminoe oyster continues to filter under similar conditions. Thus, the Suminoe oyster may be more susceptible to mortality from low dissolved oxygen conditions than the American oyster. Shell thickness is also a consideration, particularly where a species may encounter significant predation pressures, for example, from crabs. Faster growth may result in thinner shells. These factors need to be fully considered in selecting an appropriate species.
Oyster Reproduction and Growth[edit | edit source]
Oyster growth to maturity and reproduction are highly variable and are influenced by a number of factors. Reproduction is strongly associated with temperature and is also related to salinity. Predation (e.g. crabs, cow-nosed rays) is a constant threat throughout the water column, especially to larvae and young oysters. Field research and practical experience indicate that growth and mortality have strong linkages to hydrodynamics, that is, the flow of water. Other fundamental growth issues that are cross-linked with hydrodynamics include temperature, salinity, and availability of food. When structure is not available, the oysters are forced to live on the bottom where the stresses from low dissolved oxygen, sedimentation, turbidity, and salinity variations are the greatest. Under unfavorable conditions, the animals must expend more energy to survive, affecting growth rates and mortality.
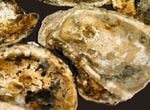
Most species, such as the American and Pacific oyster, have both male and females, although oysters change sex one or more times during their life cycle. Fertilization of eggs takes place when eggs and sperm are ejected into the surrounding water. The fertilized eggs develop into a free swimming embryo that circulates primarily with the currents. This period lasts for several weeks to a month, depending upon the species. Other species such as the European oyster possess both male and female cells. Eggs are fertilized and larvae develop inside the oyster before being ejected. Once in the water, larval oysters are vulnerable to predation, changes in temperature, salinity, etc. A spat set then occurs - surviving oyster larvae settle on and attach themselves to almost any solid object below the surface. (The life stage at which oyster larva extend a foot, settle on and cement themselves to hard substrate, is referred to as “spat set”.)
Oysters in better growing conditions grow faster than oysters in less favorable conditions. There is a greater prospect for more rapid growth off the bottom rather than on the bottom or in the sediment. In general, oysters grow faster closer in moderate to higher flows where the water around the oyster changes multiple times each hour, sedimentation is the lowest, the quality of suspended food is highest, and dissolved oxygen is the greatest. These conditions are more prevalent off the bottom, and vary in different segments of the water column. Vertical structure as substrate helps mitigate the effects of sedimentation and is more likely to put oysters where water flows are greater off the bottom. However, higher locations also increase the potential for fouling of the substrate by other encrusting marine organisms that dwell near the surface. Off-the-bottom growth rates for oysters set on Mini Bay Balls by the Chesapeake Bay Foundation and placed in Eastern Bay, a tributary to the upper Chesapeake Bay, were 2 to 3 inches in one year while oysters on the bottom in the region grow about 1 inch per year. The State Reef site that was used was in open water at the mouth of the bay with good water flow. Growth rates of 2 to 3 inches in 12 to 18 months has also been observed for off-the-bottom commercial oyster aquaculture in various locations including Maryland,Virginia, and Louisiana.
Disease[edit | edit source]
Disease is a significant threat to oysters worldwide. In order to prevent spread of infections, only disease-free seed oysters or disease free spat should be used in rehabilitation projects. Parasitic diseases such as Dermo and MSX that plague the Chesapeake Bay and the American oyster, while not toxic to humans, can kill an oyster in higher salinity waters at about the time it grows to a harvestable size (approx. 2 - 4 years depending upon growth conditions). Other parasites, such as Bonamia (which has occurred as a few locations in Europe and one in North Carolina), can very quickly devastate oyster populations in high salinity conditions.
Oysters are susceptible to other diseases. They may also ingest other pathogens and toxics that are in the water. Although production of oysters for human consumption may not be a project objective, as may be the case with a rehabilitation project, selecting a site that has the least exposure to pathogens and toxics is important. This approach gives the oysters a better chance for survival, and also reduces the potential for illness to humans if oysters are taken and eaten.
There are indications that some form of relationship may exist to an undetermined extent between infection by parasitic diseases and water depth. For example, a few private and commercial interests in the upper Chesapeake Bay have successfully grown virtually disease free oysters in floating cages at the surface. Although the life history of the parasites is uncertain, successful cage farming at the surface while oysters located at the bottom in nearby locations become infected suggests that elevating oysters higher up in the water column while keeping them sub-tidal elevation may help mitigate against this threat. However, oysters growing near the surface reach harvestable size in one to two years, while dermo and MSX parasitic infections take on the order of three years before mortality begins to occur. However, faster growth at higher elevations might increase the potential for natural oyster recruitment and growth to equal or exceed losses from parasitic diseases, absent major losses from other factors, such as harvesting.
Predation[edit | edit source]
Predation occurs throughout the water column. For example, crabs love to eat spat. Project site selection should consider the potential for localized predation, and minimize this potential insofar as practicable. Projects should minimize proximity to high value crab habitat such as seagrass beds.
Oyster Propagation[edit | edit source]
Oyster propagation basics are fundamental to rehabilitation design. Oyster propagation is normally accomplished using one or a combination of natural reproduction or hatchery assisted propagation of oyster spat (i.e., oyster larva attached to shell or a shell fragment). Our experience to date indicates that setting spat directly onto Reef Balls in a hatchery followed by interim deployment to an in-water nursery area or attaching grown-out spat on shell to Reef Balls immediately prior to deployment increases the potential for growth and survival, and thus reef development. These practices also defend against the variability of natural propagation, especially in northern climates where reproduction occurs at a lower frequency, and oysters being outcompeted by other marine organisms.
Natural Propagation[edit | edit source]
Of the two basic approaches in using designed reef structures for oyster propagation, natural propagation is the less costly but the most susceptible to highly variable natural conditions at the most critical stages. However, up to full coverage of the structure is possible, depending upon the spat set. Because there is considerable uncertainty of success when relying on natural propagation, it is desirable for the project to include multiple objectives in order to provide a return on the rehabilitation investment. The oyster rehabilitation component can be either primary or secondary objective, appropriate to the project objectives and other circumstances. Projects in which oyster rehabilitation could be a secondary benefit include shoreline stabilization projects, fishing reefs, and use of reef structures as obstructions to unauthorized harvesting activity, for example, in an oyster sanctuary.


Reef substrate or structures are placed at suitable permitted locations to await a natural spat set. If a project reef is not within the larva circulation pattern of an existing healthy oyster population of sufficient size, then the probability of success for oyster rehabilitation purposes is greatly diminished. Natural propagation is further complicated by seasonal and annual variations in the most critical environmental conditions - water temperature and salinity, and by predation, substrate fouling by other marine organisms, sedimentation, and water quality. The result is that designed reef structures may not receive a spat set or may receive a light spat set, or the spat set may occur after the structure has been fouled by other marine organisms, sedimentation, and water quality. The result is that designed reef structures may not receive a spat set or may receive a light spat set, or the spat set may occur after the structure has been fouled by other marine organisms. Although a light spat set could be beneficial to the biological and reef structure value of the immediate reef structure, the contribution to oyster population rehabilitation objectives would be minimal at best, and potentially not self-sustaining.
Hatchery-Assisted Propagation[edit | edit source]
Hatchery techniques used with design reef structures include:
- Setting oyster spat directly onto small designed reef structures.
- Setting oyster spat directly onto small designed reef structures with shell fragments or shell embedded into outer layers.
- Setting oyster spat onto plugs for later attachment to a designed reef structure.
- Setting oyster spat onto shell in spat bags for later attachment to a designed reef structure.

Hatchery propagation involves simulation of natural conditions that induce oyster broodstock to spawn. Only disease-free broodstock should be used. Oyster larvae are released into large tanks where they settle on the substrate, typically spat bags full of oyster shell. The spat on shell is then taken from the tanks and placed temporarily into grow-out areas to gain size before deployment to an oyster bed or reef. Somewhat larger sizes makes the juvenile oysters less vulnerable to predation. The timing and duration of hatchery services including grow-out need to be factored into reef project development schedule.
Under identical conditions with multiple tanks, spat set will vary by tank, who knows why. The substrate is removed and the spat set is manually counted as a baseline for future performance monitoring. The substrate is removed from the hatchery tanks to an in-water nursery area or immediately transported for deployment. Although data are limited, grow out in a low wave energy protected location until the juvenile oysters are well established has yielded more favorable results than immediate deployment to reef site. The substrate with oysters attached is then deployed at the reef site.
If plugs or spat on shell are used, the material is attached to the designed reef structure using a non-toxic compound, typically an epoxy that cures underwater. This may be used as the primary method or to supplement spat set directly onto a reef structure. If the former, then only partial coverage of the structure is probable because the spaced not used is likely to be fouled by other marine species unless the structure receives a natural spat set shortly after deployment. One project to date that used plugs indicated that coral plug adapters made from the concrete mix was marginal for oysters. Setting spat directly to the structures or to oyster shell (for later attachment to the structures) yielded a better result during the same field experiment. A difficulty with plugs was that some spat set on the sides, complication attachment. A plug composed largely of shell fragments might potentially achieve better spat sets, but this concept was not tested.
Shell can be added to Reef Balls in several ways to improve the surface for spat set at the hatchery (as well as for natural propagation). One technique is to use aggregate that already includes shell fragments, as is often the case in Florida. Another technique is to fold oyster shell fragments into the concrete mix along the sides of the mold as the concrete is being poured into the Reef Ball mold. Natural oyster shell is more durable than clam shell and is a better choice. However, only a portion of the shell fragments will occur at the exposed surface. A more time consuming but more reliable approach is to mold oyster shell onto the Reef Ball surface during construction. This can be accomplished by manually oyster shells against the side of the mold, with the interior of the shell facing outward against the interior of the mold. This technique relies on the rugged shape of the exterior side of the oyster shell to provide sufficient protrusions so as to anchor the shell in the concrete.

An alternative method that reduces the potential for shell to dislodge is to manually attach an anchor point that protrudes from the backside of the shell into the concrete to insure a permanent attachment. This can be accomplished by attaching a Reef Ball coral plug adaptor to the natural exterior side of the oyster shell. The smaller diameter end of the coral plug adaptor can be attached to the shell using non-toxic marine epoxy. The shells are then manually placed in successive layers along the interior of the mold with the coral plug adaptor closest to the center of the mold. As the concrete is slowly poured in, it forms around the adaptor, making the attachment permanent. If a shell with spat were to fall off during transit, it can be reattached with non-toxic marine epoxy just prior to deployment at the reef site. Should a shell with spat fall off once deployed, the spat may still survive in its natural benthic habitat.
Physical Conditions[edit | edit source]
A fundamental question is whether or not a prospective rehabilitation site can actually support oyster growth. Site hydrodynamics linkages to the distribution of food and larvae, water quality oxygen, salinity, temperature, and sedimentation are important. For a site where harvesting may be allowed once oysters reach maturity, whether or not the site would produce oysters suitable for eating would also be an issue. For example, oysters may be able to survive in higher concentrations of pathogens and toxics than might be allowed for shellfish harvesting.
Water Circulation - Distribution of Food and Larvae[edit | edit source]
The circulation of water is a fundamental issue because oysters are in a fixed location, and completely dependent upon the currents to bring them food and circulate larvae. Assuming that continuing natural recruitment to a reef site is a project objective, the reef site must be in the distribution pattern for naturally produced larvae. Hydrodynamic and biological assessments are fundamental to determining whether or not this condition will exist. If not, then the investment in the reef might still produce nice fish habitat, but not a return on investment relative to restoring oyster populations. Considering the resources and time required for the necessary studies, designed reef structures with oyster rehabilitation as a primary objective should be correlated with existing studies and oyster rehabilitation activities to the maximum extent that is practicable.
Water Depth[edit | edit source]
In addition to water depth issues discussed earlier, oysters (and reef structures use for oyster rehabilitation) must be within the biological tide line for the selected species. Maintaining subtidal but off-the-bottom growing conditions appears to improve growth potential as discussed earlier, the fact that intertidal oyster species such as the American oyster can remain out of the water for one tidal cycle. However, when out of the water, the oysters clam up, so to speak. They obviously cannot filter feed under such conditions. Although subtidal oysters can survive being frozen while in the water, oysters freeze when exposed to subfreezing air temperatures, often resulting in mortality.
Water Quality[edit | edit source]
Oysters in large quantities in relation to the size of the water body can improve water quality. However, water quality must be good enough for oysters to survive and grow to maturity. The better the water quality, the lesser the stress on the oyster. If water quality at a proposed project site is toxic to oysters or could become so seasonally, then the project site, or lower portions of the water column in the case of low dissolved oxygen levels, may not be suitable for some or all species.
Salinity Range[edit | edit source]
The salinity in an estuary varies seasonally and annually, depending upon the inflow of fresh water from rivers, streams, and surrounding land masses. The multi-year salinity range at the project site must be within the salinity tolerances of the selected oyster species. Oyster growth and reproduction are related to salinity and each other, as well as to temperature. Spat sets will vary from none to heavy, depending upon the correlation of these conditions with oyster biology. This is critically important when relying on a natural spat set to colonize a structure. If a spat set does not occur while the structure is fresh, it will become fouled by other marine organisms and most likely preclude future oyster recruitment.
Oysters reproduce when certain salinity thresholds are crossed, either up or down, depending upon the species. For example, the American and Suminoe oyster require approximately the same conditions for spawning. However, on a relative scale, the American oyster requires an increase above a salinity threshold whereas the Suminoe oyster requires a decrease below a similar threshold. When the Suminoe oyster is in higher salinity water, it does not reproduce. All energy is directed to growth, resulting in accelerated growth. In contrast, the American oyster grows slower in less saline waters but requires more salty conditions for reproduction where it is more susceptible to parasitic diseases which also need more salty conditions.
Water Temperature[edit | edit source]
Oyster growth and reproduction are related to temperature. The temperature range at the project site must be within the temperature tolerances of the oyster species that is selected. The temperatures are which oyster reproduce varies by species. In general, northern species spawn at somewhat lower temperatures than southern oyster species. Although feeding habitats vary by species, oysters are more active in warmer water. Thus, oysters would be expected to grow at faster rates in warmer waters, all other factors being equal. This affects the time required for reef development, and needs to be considering in establishing project monitoring and evaluation criteria.
Vertical Structure[edit | edit source]
When structure is not available, oysters live at the bottom where the stresses from low dissolved oxygen, sedimentation, turbidity, and salinity variations are the greatest. In some areas such as the Chesapeake Bay, natural structure may not be present or greatly diminished relative to historical conditions. Structural characteristics of historic oyster reefs, sometimes referred to as “oyster rock,” may have been reduced or removed through harvesting methods. For example, much of the oyster reefs in the Chesapeake Bay were virtually leveled by mechanical harvesting over a hundred years ago. Or, effects of storm waves or tsunamis may have displaced or covered natural structure.
Reef structures should be designed not only get oysters off the bottom and into the water flow, but also provide sufficient vertical elevation to provide many years of use relative to sedimentation rates. Reef elevation also needs to specifically address overhead clearances to avoid becoming an obstruction to navigation, unless lesser overhead depths are capable of being permitted. In the latter case, obstruction markers will be normally be required as a permit condition.
Sedimentation[edit | edit source]
Site selection must minimize the potential for reef structures to be buried through the accretion of sediment. Locations selected for oyster reefs should have low sedimentation rates or alternately, sedimentation rates that do not exceed the rate of natural increase in elevation through oyster growth and recruitment of spat to the reef structure. An exception is beach stabilization structures where accretion of areas shoreward of the reef is usually a project objective. In this case, rehabilitation of oyster habitat would necessarily be a secondary objective with shoreline or beach stabilization taking precedence. Survival of oysters on such reefs depends on whether or not the oysters get buried. Setting spat directly to reef modules in a hatchery for use at a shoreline stabilization project is not recommended if sediment accretion is expected. If spat are to be attached to the reef structures, then the shell or other substrate to which the spat is attached should, in general, be placed on the upper portions of the reef structure and on those that are farthest away from the shoreline where the water would normally be the deepest.
Substrate[edit | edit source]
Oyster larvae have a strong preference for oyster shell as substrate, although they will attach to almost any hard substrate. Oyster shell generally provides consistent results in hatcheries. Where oyster shell has not been available or not available in sufficient quantity or is too expensive, alternative materials have been used as a foundation for reefs for either natural spat set or deployment of spat on shell, and as substrate for hatchery operations. Options include natural material (e.g., clam shell, conch shell, granite, limestone marl), demolition materials (e.g. weathered concrete rubble free of other debris), and industrial byproducts (e.g., slag with limestone content), and manufactured materials (e.g. tiles).
When using designed reef structures as substrate, bottom conditions must be capable of supporting the structure. If bottom conditions are not favorable, it may be possible to establish a foundation using shell, weathered concrete rubble, or other suitable materials. In view of the costs involved, the suggested approach for small-scale projects is to only use sites that are suitable without modification, or to piggyback the reef structure with another project that establishes a suitable foundation.
Designed reef structure should be cured to their applicable technical specifications prior to deployment. Weathering ashore exposed to the elements for a few months prior to deployment can help “season” the surface and allows further curing to build additional strength. Additional in-water seasoning for a short period, on the order of a few weeks, allows interaction between the surface and ambient conditions, making it appear more natural to oyster larvae. In any case, the modules need to be deployed just prior to the projected start of annual oyster spawning in order to have the best chance of catching a natural spat set while lessening the potential for biofouling by other marine species.
Surface Texture[edit | edit source]

A very rough surface is highly desirable. Nooks, crannies, and crevices provide both attachment points and cover for oyster larvae. Oysters have also been observed on smooth sided artificial ree structures. In one location in Maryland’s Eastern Bay, unusually formed American oysters were observed on smooth-sided reef structures. Uniquely, the oysters were relative flat and one sided; they used the smooth side of the structure as part of their enclosure. Thus, the animal would be killed if it were removed. It is not known whether or not this was a one-time phenomenon. However, it is known that the American oyster performs well in the presence of highly variable surface conditions.
Reef Horizontal Configuration[edit | edit source]

The importance of reef horizontal configuration relative to the oyster life cycle is uncertain. Reef configuration is related to hydrodynamic conditions. When project objectives include rehabilitation of reef structure, attempting to place structures so as to resemble natural formations and associated hydrodynamic effects and circulation patterns is a logical approach – if the number of modules and foundation conditions are sufficient. Local area data may be available to assist with determining historical reef configurations. If not, then configuring the structures somewhat across the predominant line of flow could increase mixing of the water around the structures, improving distribution of food and potentially increasing dissolved oxygen.
Deployment[edit | edit source]
Deployment of spat and reef structures with set or attached spat must be carefully planned and must be within the oyster species biological tolerances. The capability of oysters to remain out of the water for one tidal cycle is critically important to transporting and deploying oyster spat and oyster stock to beds and reefs. This is a time consuming process, the duration of which depends upon distance to the oyster bed or reef, vessel transit speeds and deployment capabilities, and other transportation factors. However, it may be possible to “push” the time oysters can remain out of the water by keeping them wet using water drawn from the estuary. It is also important to keep the oysters from being “cooked” while they or structures with oysters attached are on deck. For example, a tarp could be placed above, but not on, the reef structures or oyster spat, thereby shading the animals but allowing air circulation.
If oyster spat on shell or other suitable substrate is going to be manually attached to reef structures, this should be done just prior to structure placement in the water in order to minimize the potential for mortality on the structures surface, for example, from heating by the Sun. Spat on shell can be attached using non-toxic marine epoxy that cures underwater. For Reef Balls, the shells with spat can be attached to tops of the modules around the center holes as well as the bottom half of the side holes. Smaller shells with spat can be attached to the upper third to half of the Reef Ball surface. Attachment below the middle of the Reef Ball is more susceptible to falling off before the epoxy cures sufficiently to hold the shell permanently in plan.

The designed reef structures should be moved gently and placed on the bottom so as to minimize the risk of damage to the modules and attached spat. When using marine heavy lifts, recognize that this approach is dangerous. Qualified personnel and all appropriate safety precautions should be used to protect individuals equipment, designed reef structures, and attached animals. Fabric lifting straps with adequate rated lifting capacity, preferably with integrated chafing protection, work very well for lifting and deploying Reef Balls and minimize the potential for damage. Marine hooks of sufficient rated capacity with manually controlled or automatic bottom release capability have also worked very well for Reef Ball deployment. Other equipment, lifting slings, and hooks have been used successfully by qualified marine personnel. Floating deployment may also be an option in some locations. The technique is the same that as applied for deploying Reef Balls for coral reef rehabilitation.
Monitoring[edit | edit source]

All projects should include environmental monitoring to ascertain performance relative to project specifications and objectives. This is appropriate not only to assess performance, but also to gain lessons learned and to develop data to support planning of subsequent projects. Monitoring should establish a baseline condition of deployed structures as well as initial reef profiles. Subsequent monitoring should determine and assess reef biological performance. Underwater photography using divers or photographic equipment deployed from the surface have been used effectively. Bay Balls and Mini Bay Balls at several locations in the upper Chesapeake Bay were lifted from the bottom for inspection on deck where close inspection.
Disclaimer[edit | edit source]
This information was Reef Ball's Draftguide document.
References[edit | edit source]
A substantial body of literature exists for oyster cultivation and rehabilitation. Oyster information can be readily accessed through the worldwide web. U.S. Government sources include NOAA’s Aquaculture Information Center. Suggested keywords include: oyster; oyster aquaculture, beds, farming, parasites, predation, propagation, reefs, rehabilitation; American oyster, Asian oyster, European oyster, Pacific oyster, Suminoe oyster; Crassostrea virginica, Crassostrea ariakensis, Crassostrea gigas, Ostrea edulis; dermo, MSX.
Suggested reading:
- Chesapeake Research Consortium. June, 1999. Chesapeake Bay Oyster Restoration: Consensus of a Meeting of Scientific Experts, Virginia Institute of Marine Science, Wachapreague, Virginia. Gloucester Point, VA: Virginia Institute of Marine Science. www.vims.edu
- Goldsborough, William, and Donald Meritt. 2001. Oyster Gardening for Restoration and Education. Publication Number UM-SG-SGEP-99-05. College Park, Md.: Maryland Sea Grant College Program.www.mdsg.umd.edu/issues/chesapeake/oysters/garden/index.html Kemp, Philip S., ed. November, 2005. Oyster Gardening Manual: An environmental demonstration project for North Carolina. Carteret Community College, Aquaculture Tech Program. [main2.carteret.edu/~kemps/files/Oyster%20Gardening%20Manual%20print.pdf]
- Maryland Environmental Service (MES). August, 2006. Reef Ball Monitoring and Quality Assessment Report. Millersville, Md.: MES. www.reefball.org
- McKay, Laura, and Virginia Witmer. 2006. Virginia Oyster Gardening. Virginia Coastal Zone Management Program. www.deq.virginia.gov/coastal/documents/
- Oyster Diseases of the Chesapeake Bay. n.d. Gloucester Point, VA: Virginia Institute of Marine Science. www.vims.edu/newsmedia/pdfs/oyster-diseases-CB.pdf
- Oyster Farming in Louisiana. n.d. Louisiana Sea Grant College Program. www.lamer.lsu.edu/classroom/edonahalfshell/pdf/cycle_info.pdf
- Takacs, Rich, Jamie King, and Paula Jasinski. 2005. Oyster Restoration on the East and Gulf Coasts. Habitat Connections 5:2. National Oceanic and Atmospheric Administration. [chesapeakebay.noaa.gov/docs/HabitatConnectVol5no2May2005.pdf]
- Wallace, Richard K. August, 2001. Cultivating the Eastern Oyster, Crassostrea virginica. SRAC Publication No. 42. Southern Regional Aquaculture Center. [www.agmrc.org/agmrc/commodity/aquaculture/oysters/ AGMRC]