(From The Household Cyclopedia, 1881. This article contains antique processes, some of which are dangerous.)
Expeditious Mode of Reducing Iron Ore into Malleable Iron.[edit | edit source]
The way of proceeding is by stamping, washing, etc., the calcine and materials, to separate the ore from extraneous matter; then fusing the prepared ore in an open furnace, and instead of casting it, to suffer it to remain at the bottom of the furnace till it becomes cold.
New Method of Shingling and Manufacturing Iron.[edit | edit source]
The ore being fused in a reverberating furnace, is conveyed, while fluid, into an air-furnace, where it is exposed to a strong heat till a bluish flame is observed on the surface, it is then agitated on the surface till it loses its fusibility and is collected into lumps galled loops. These loops are then put into another air-furnace, brought to a white or welding heat, and then shingled into halfblooms or slabes. They are again exposed to the airfurnace, and the half-blooms taken out and forged into anconies, bars, halfflats, and rods for wire; while the slabes are passed, when of a welding heat, through the grooved rollers. In this way of proceeding, it matters not whether the iron is prepared from cold or hot short metal, nor is there any occasion for the use of finery, charcoal, coke, chafery or hollow-fire; or any blast by bellows or otherwise, or the use of fluxes in any part of the process.
Approved Method of Welding Iron.[edit | edit source]
This consists in the skilful bundling of the iron to be welded, in the use of an extraordinarily large forgehammer, in employing a bulling-furnace, instead of a hollowfire or chafery, and in passing the iron, reduced to a melting heat, through grooved mill rollers of different shapes and sizes, as required.
Welding Steel, or Iron and Cast Steel.[edit | edit source]
Melt borax in an earthen vessel, and add 1-10th of pounded sal ammoniac. When well mixed, pour it out on an iron plate, and as soon as it is cold, pulverize and mix it with an equal quantity of unslaked lime. To proceed to the operation, the iron or steel must be first heated to a red heat, and the powder strewed over it; the pieces of metal thus prepared are to be again put in the fire, and raised to a heat considerably;ower than the usual welding one, when it is to be withdrawn and well beaten by a hammer till the surfaces are perfectly united.
Welding by Pressure.[edit | edit source]
Soft metals can be welded cold by great pressure and recently hydraulic pressure has been applied by M. Duportail to the welding of heated masses of iron. The advantage of pressure over hammering, is that it reaches the centre of the bar and produces a homogeneous weld.
Common Hardening.[edit | edit source]
Iron by being heated red-hot, and plunged into cold water, acquires a great degree of hardness. This proceeds from the coldness of the water which contracts the particles of the iron into less space.
Case-hardening.[edit | edit source]
Case-hardening is a superficial conversion of iron into steel by cementation. It is performed on small pieces of iron by enclosing them in an iron box containing burnt leather, bone-dust, or ferrocyanide of potassium, and exposing them for some hours to a red heat. The surface of the iron thus becomes perfectly hardened. Iron thus treated is susceptible of the finest polish.
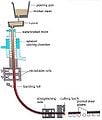
To Convert Iron into Steel by Cementation.[edit | edit source]
The iron is formed into bars of a convenient size, and then placed in a cementing furnace with a sufficient quantity of cement, which is composed of coals of animal or vegetable substances, mixed with calcined bones, etc. The following are excellent cements: 1st, 1 part of powdered charcoal and 1/2 a part of wood-ashes well mixed together; or, 2nd, 2 parts of charcoal, moderately powdered, 1 part of borax, horn, hair, or skins of animals, burnt in close vessels to blackness, and powdered, and 1/2 a part of wood-ashes; mix them well together. The bars of iron converted into steel, are placed upon a stratum of cement, and covered all over with the same, and the vessel which contains them, closely luted, must be exposed to a red heat for 8 or 10 hours, when the iron will be converted into steel.
Steel is prepared from bar-iron by fusion; which consists of plunging a bar into melted iron, and keeping it there for some time, by which process it is converted into good steel.
All iron which becomes harder by suddenly quenching in cold water is called steel; and that steel which in quenching acquires the greatest degree of hardness in the lowest degree of heat, and retains the greatest strength in and after induration, ought to be considered as the best.
Improved Process of Hardening Steel[edit | edit source]
Articles manufactured of steel for the purposes of cutting, are, almost without an exception, hardened from the anvil; in other words, they are taken from the forger to the hardener without undergoing any intermediate process; and such is the accustomed routine, that the mischief arising has escaped observation. The act of forging produces a strong scale or coating, which is spread over the whole of the blade; and to make the evil still more formidable, this scale or coating is unequal in substance, varying in proportion to the degree of heat communicated to the steel in forging: it is, partially, almost impenetrable to the action of water when immersed for the purpose of hardening. Hence it is that different degrees of hardness prevail in nearly every razor manufactured; this is evidently a positive defect, and so long as it continues to exist, great difference of temperature must exist likewise. Razor-blades not unfrequently exhibit the fact here stated in a very striking manner; what are termed clouds, or parts of unequal polish, derive their origin from this cause; and clearly and distinctly, or rather distinctly though not clearly, show how far this partial coating has extended, and where the action of the water has been yielded to, and where resisted.
It certainly cannot be matter of astonishment, that so few improvements hove been made in the hardening of steel, when the evil here complained of so universally obtains, as almost to warrant the supposition that no attempt has ever been made to remove it. The remedy, however, is easy and simple in the extreme, and so evidently effi-cient in its application, that it cannot but excite surprise, that, in the present highly improved state of our manufactures, such a communication should be made as a discovery entirely new.
Instead, therefore, of the customary mode of hardening the blade from the anvil, let it be passed immediately from the hands of the forger to the grinder; a slight application of the stone will remove the whole of the scale or coating, and the razor will then be properly prepared to undergo the operation of hardening with advantage. It will be easily ascertained, that steel in this state heats in the fire with greater regularity, and that when immersed, the obstacles being removed to the immediate action of the water on the body of the steel, the latter becomes equally hard from one extremity to the other. To this may be added that, as the lowest possible heat at which steel becomes hard is indubitably the best, the mode here recommended will be found the only one by which the process of hardening can be effected with a less portion of fire than is, or can be, required in any other way. These observations are decisive, and will, in all probability, tend to establish in general use, what cannot but be regarded as a very important improvement in the manufacturing of edged steel instruments. Rhodes’ Essay on the Manufacture of a Razor.
Improved Mode of Hardening Steel by Hammering Gravers, axes, and in fact all steel instruments that require to be excessively hard, may be easily rendered so by heating them to the tempering degree and hammering them till cold. If a graver, it is to be heated to a straw-color, hammered on the acute edge of the belly, tempered to the straw color again, ground and whetted to a proper shape. A graver thus prepared will cut into steel, without previous decarbonization. If the point should on trim be found not sufficiently hard, the operation of heating, hammering, and tempering, etc., may be repeated as often as necessary.
English Cast-Steel.[edit | edit source]
The finest of steel, called English cast-steel, is prepared by breaking to pieces blistered steel, and then melting it in a crucible with a flux composed of carbonaceous and vitrifiable ingredients. The vitrifiable ingredient is used only inasmuch as it is a fusible body, which flows over the surface of the metal in the crucibles, and prevents the access of the oxygen of the atmosphere. Broken glass is sometimes used for this purpose.
When thoroughly fused it is cast into ingots, which. by gentle heating and careful hammering, are tilted into bars. By this process the steel becomes more highly carbonized in proportion to the quantity of flux, and in consequence is more brittle and fusible than before. Hence it surpasses all other steel in uniformity of texture, hardness, and closeness of grain, and is the material employed in all the finest articles of English cutlery.
To make Edge-tools from Cast-Steel and Iron.[edit | edit source]
This method consists in fixing a clean piece of wrought iron, brought to a welding-heat, in the centre of a mould, and then pouring in melted steel, so as entirely to envelop the iron; and then forging the mass into the shape required.
To Color Steel Blue.[edit | edit source]
The steel must be finely polished on its surface, and then exposed to a uniform degree of heat. Accordingly, there are three ways of coloring: first, by a flame producing no soot, as spirit of wine; secondly, by a hot plate of iron; and thirdly, by wood-ashes. As a very regular degree of heat is necessary, wood-ashes for fine work bears the preference. The work must be covered over with them, and carefully watched; when the color is suffi-ciently heightened, the work is perfect. This color is occasionally taken off with a very dilute muriatic acid.
To Distinguish Steel from Iron.[edit | edit source]
The principal characters by which steel may be distinguished from iron, are as follows:-
1. After being polished, steel appears of a whiter light gray hue, without the blue cast exhibited by iron. It also takes a higher polish.
2. The hardest steel, when not annealed, appears granulated, but dull, and without shining fibres.
3. When steeped in acids the harder the steel is, of a darker hue is its surface.
4. Steel is not so much inclined to rust as iron.
5. In general, steel has a greater specific gravity.
6. By being hardened and wrought, it may be rendered much more elastic than iron.
7. It is not attracted so strongly by the magnet as soft iron. It likewise acquires magnetic properties more slowly, but retains them longer; for which reason, steel is used in making needles for compasses and artificial magnets.
8. Steel is ignited sooner, and fuses with less degree of heat than malleable iron, which can scarcely be made to fuse without the addition of powdered charcoal; by which it is converted into steel, and afterwards into crude iron.
9. Polished steel is sooner tinged by heat, and that with higher colors than iron.
10. In a calcining heat, it suffers less loss by burning than soft iron does in the same heat, and the same time. In calcination a light blue flame hovers over the steel, either with or without a sulphurous odor.
11. The scales of steel are harder and sharper than those of iron and consequently more fit for polishing with.
12. In a white heat, when exposed to the blast of the bellows among the coals, it begins to sweat wet, or melt, partly with light-colored and bright and partly with red sparkles, but less crackling than those of iron. In a melting heat, too, it consumes faster.
13. In the sulphuric, nitric, and other acids steel is violently attacked, but is longer in dissolving than iron. After maceration, according as it is softer or harder, it appears of a lighter or darker gray color; while iron on the other hand is white.
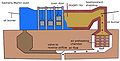
The Bessemer Process of Making Steel.[edit | edit source]
Hematite pig-iron smelted with coke and hot-blast has chiefly been used. Themetal is melted in a reverberatory furnace, and is then run into a founder’s ladle, and from thence it is transferred to the vessel in which its conversion into steel is to be effected. It is made of stout plate iron and lined with a powdered argillaceous stone found in this neighborhood below the coal, and known as ganister. The converting vessel is mounted on axes, which rest on stout iron standards, and by means of a wheel and handle it may be turned into any required position. There is an opening at the top for the inlet and pouring out of the metal, and at the lowest part are inserted 7 fire-clay tuyeres, each having five openings in them; these openings communicate at one end with the interior of the vessel, and at the other end with a box called the tuyere-box, into which a current of air from a suitable blast engine is conveyed under a pressure of about 14 lbs. to the square inch, a pressure more than sufficient to prevent the fluid metal from entering the tuyeres. Before commencing the first operation, the interior of the vessel is heated by coke, a blast through the tuyeres being used to urge the fire. When sufficiently heated, the vessel is turned upside down and all the unburned coke is shaken out. The molten pig-iron is then run in from the ladle before referred to; the vessel, during the pouring in of the iron, is kept in such a position that the ori-fices of the tuyeres are at a higher level than the surface of the metal. When all the iron has run in the blast is turned on, and the vessel quickly moved round. The air then rushes upwards into fluid metal from each of the 35 small orifices of the tuyeres, producing a most violent agitation of the whole mass. The silicium, always present in greater or less quantities in pig-iron, is first attacked. It unites readily with the oxygen of the air, producing silicic acid; at the same time a small portion of the iron undergoes oxidation, hence a fluid silicate of the oxide of iron is formed, a little carbon being simultaneously eliminated. The heat is thus gradually increased until nearly the whole of the silicium is oxidized; this generally takes place in about 12 minutes from the commencement of the process. The carbon now begins to unite more freely with the oxygen of the air, producing at first a small flame, which rapidly increases, and in about three minutes from its first appearance we have a most intense combustion going on: the metal rises higher and higher in the vessel, sometimes occupying more than double its former space. The frothy liquid now presents an enormous surface to the action of the oxygen of the air, which unites rapidly with the carbon contained in the crude iron, and produces a most intense combustion, the whole, in fact, being a perfect mixture of metal and fire. The carbon is now eliminated so rapidly as to produce a series of harmless explosions, throwing out the fluid slags in great quantities while the union of the gases is so perfect that a voluminous white flame rushes from the mouth of the vessel, illuminating the whole building, and indicating to the practiced eye the precise condition of the metal inside. The workman may thus leave off whenever the number of minutes he has been blowing and the appearance of the flame indicate the required quality of the metal. This is the mode preferred in working the process in Sweden. But here we prefer to blow the metal until the flame suddenly stops, which it does just on the approach of the metal to the condition of malleable iron: a small quantity of charcoal pigiron, containing a known quantity of carbon, is then added, and steel is produced of any desired degree of carburation, the process having occupied about 28 minutes from the commencement. The vessel is then turned, and the fluid steel is run into the casting ladle, which is provided with a plug rod covered with loam: the rod posses over the top of the ladle, and works in guides on the outside of it, so that, by means of a lever handle, the workmen may move it up and down as desired. The lower part of the plug, which occupies the interior of the ladle, has fitted to its lower end a fireclay cone, which rests in a seating of the same material let into the bottom of the ladle, thus forming a cone valve, by means of which the fluid steel is run into different-sized moulds, as may be required, the stream of fluid steel being prevented by the valve plug from flowing during the movement of the casting ladle from one mould to another. By tapping the metal from below, no scoria or other extraneous floating matters are allowed to pass into the mould.
Uchatius Steel.[edit | edit source]
Pig iron is first granulated by running it in a small stream into cold water kept constantly agitated. The granulated metal is mixed with sparry iron ore, and if necessary a small portion of manganese, and heated in crucibles in the ordinary cast-steel blast furnace.
Gallery[edit | edit source]
-
Siemens Martin furnace
-
Hot profile rolling
-
LD-process